Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.22 Uncertainty in volumetric analysis-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.22 Uncertainty in volumetric analysis- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.22 Uncertainty in volumetric analysis- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.22 understand how to minimise the sources of measurement uncertainty in volumetric analysis and estimate the overall uncertainty in the calculated result
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.22 Minimising Uncertainty in Volumetric Analysis & Estimating Overall Uncertainty
Volumetric analysis (titration) involves measurement uncertainties from apparatus and technique. These must be minimised and quantified to assess reliability of results.
(A) Sources of Uncertainty 
- Burette readings (±0.05 cm\(^3\))
- Pipette volume (e.g. ±0.06 cm\(^3\))
- End-point judgement (indicator colour change)
- Parallax error when reading meniscus
(B) Minimising Uncertainty
1. Technique Improvements
- Read meniscus at eye level
- Use white tile to see colour change clearly
- Add solution dropwise near endpoint
- Swirl continuously for complete mixing
2. Repeating Measurements
- Perform multiple titrations
- Obtain concordant titres (within ±0.10 cm\(^3\))
- Take average of concordant values
3. Apparatus Handling
- Rinse burette with solution used
- Remove air bubbles from burette tip
- Use volumetric pipette for accuracy
(C) Calculating Percentage Uncertainty
For a single measurement:
\( \mathrm{\%\ uncertainty = \frac{absolute\ uncertainty}{measured\ value} \times 100} \)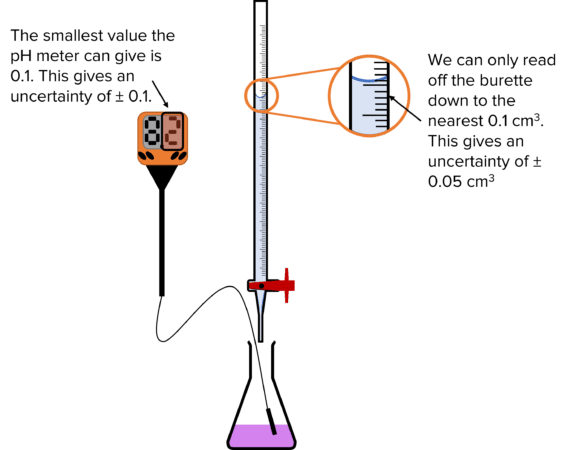
Burette Example
- Two readings (initial + final) → total uncertainty = ±0.10 cm\(^3\)
\( \mathrm{\%\ uncertainty = \frac{0.10}{titre} \times 100} \)
(D) Total Uncertainty in Result
- Add percentage uncertainties from:
- Burette
- Pipette
Rule:
Total % uncertainty ≈ sum of individual % uncertainties
(E) Key Insight
- Larger titre → smaller % uncertainty
- Small volumes → larger % error
Summary
- Minimise errors using careful technique and repetition.
- Calculate % uncertainty for each measurement.
- Add uncertainties to estimate total error.
Therefore, reliable titration results depend on both precision and proper uncertainty analysis.
Example 1
A titration uses a burette (±0.05 cm\(^3\) per reading) and a 25.0 cm\(^3\) pipette (±0.06 cm\(^3\)). The average titre is 24.80 cm\(^3\). Calculate the total percentage uncertainty in the result.
▶️ Answer/Explanation
Burette uncertainty:
Two readings → \( \mathrm{±0.10\ cm^3} \)
\( \mathrm{\%\ = \frac{0.10}{24.80} \times 100 = 0.40\%} \)
Pipette uncertainty:
\( \mathrm{\%\ = \frac{0.06}{25.0} \times 100 = 0.24\%} \)
Total uncertainty:
\( \mathrm{0.40 + 0.24 = 0.64\%} \)
Final Answer: \( \mathrm{0.64\%} \)
Example 2
A student obtains a titre of 10.20 cm\(^3\) in a titration. Explain why this result has a higher percentage uncertainty than a titre of 25.00 cm\(^3\), and suggest two improvements.
▶️ Answer/Explanation
Explanation:
Percentage uncertainty depends on measured volume.
Smaller titre → same absolute error is a larger percentage.
\( \mathrm{\%\ uncertainty = \frac{0.10}{10.20} \times 100 \approx 0.98\%} \)
This is higher than for 25.00 cm\(^3\).
Improvements:
- Use a more dilute solution → increases titre volume
- Repeat titration and average concordant results
