Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.23 CORE PRACTICAL 4: Standard solution-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.23 CORE PRACTICAL 4: Standard solution- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.23 CORE PRACTICAL 4: Standard solution- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.23 CORE PRACTICAL 4
Preparation of a standard solution from a solid acid and use it to find the concentration of a solution of sodium hydroxide
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.23 CORE PRACTICAL 4 – Preparation of a Standard Solution & Titration with NaOH
This practical involves:
- Preparing a standard solution from a solid acid (e.g. oxalic acid)
- Using it to determine the concentration of \( \mathrm{NaOH} \) by titration
(A) Preparation of Standard Solution
Procedure
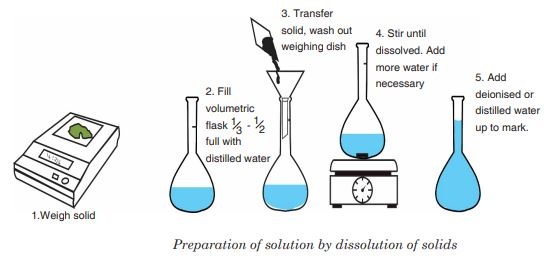
- Accurately weigh solid acid (e.g. \( \mathrm{H_2C_2O_4 \cdot 2H_2O} \))
- Transfer to a beaker and dissolve in distilled water
- Transfer solution into a volumetric flask
- Rinse beaker and add washings to flask
- Make up to the mark with distilled water
- Stopper and invert several times to mix
Key Equation
\( \mathrm{n = \frac{mass}{M_r}} \), then \( \mathrm{c = \frac{n}{V}} \)
(B) Titration with Sodium Hydroxide
Procedure
- Pipette 25.0 cm\(^3\) of standard acid into conical flask
- Add phenolphthalein indicator
- Fill burette with \( \mathrm{NaOH} \)
- Titrate until solution turns pale pink
- Record titre and repeat for concordant values
Indicator Choice
- Phenolphthalein: colourless → pink
- Suitable for weak acid–strong base titration
(C) Calculations
- Step 1: Calculate moles of acid
- Step 2: Use mole ratio
- Step 3: Find moles of \( \mathrm{NaOH} \)
- Step 4: Calculate concentration of \( \mathrm{NaOH} \)
Key Skills
- Accurate weighing (analytical balance)
- Quantitative transfer (washing technique)
- Reading meniscus correctly
- Obtaining concordant titres
Sources of Error
- Loss of solid during transfer
- Overshooting endpoint
- Incomplete mixing
Summary
- Prepare standard solution accurately from solid acid.
- Use it to determine unknown \( \mathrm{NaOH} \) concentration.
- Based on stoichiometry and titration technique.
Therefore, this practical combines preparation accuracy with titration analysis.
Example 1
1.26 g of \( \mathrm{H_2C_2O_4 \cdot 2H_2O} \) is dissolved to make 250 cm\(^3\) solution. 25.0 cm\(^3\) requires 20.0 cm\(^3\) NaOH. Find concentration of NaOH.
▶️ Answer/Explanation
\( M_r = 126 \)
\( \mathrm{n = \frac{1.26}{126} = 0.0100\ mol} \)
Volume = 0.250 dm\(^3\)
\( \mathrm{c = \frac{0.0100}{0.250} = 0.0400\ mol\ dm^{-3}} \)
Moles in 25.0 cm\(^3\):
\( \mathrm{0.0400 \times 0.0250 = 0.00100\ mol} \)
Ratio 1:2 → NaOH = 0.00200 mol
\( \mathrm{c = \frac{0.00200}{0.0200} = 0.100\ mol\ dm^{-3}} \)
Example 2
Explain why it is important to rinse the volumetric flask with distilled water after transferring the solution.
▶️ Answer/Explanation
Ensures all solute is transferred into the flask.
Prevents loss of substance.
Maintains accurate concentration.
