Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.24 Trends (mp, bp, electronegativity, reactivity)-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.24 Trends (mp, bp, electronegativity, reactivity)- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.24 Trends (mp, bp, electronegativity, reactivity)- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.24 understand reasons for the trends for Group 7 elements in:
i melting and boiling temperatures and physical state at room temperature
ii electronegativity
iii reactivity down the group
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.24 Trends in Group 7 (Halogens)
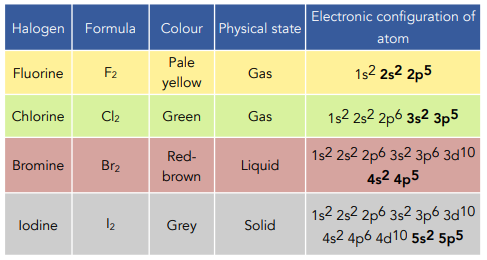
Group 7 elements (halogens) include \( \mathrm{F_2, Cl_2, Br_2, I_2} \). Their properties show clear trends down the group due to changes in atomic size, intermolecular forces, and electron attraction.
(i) Melting & Boiling Points + Physical State
Trend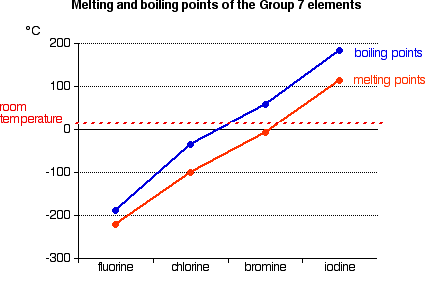
- Melting and boiling points increase down the group.
- Physical state changes:
- \( \mathrm{F_2, Cl_2} \) → gases
- \( \mathrm{Br_2} \) → liquid
- \( \mathrm{I_2} \) → solid
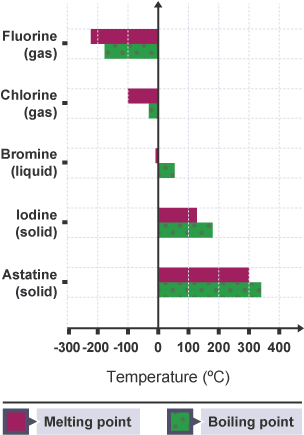
Explanation
- Halogens exist as diatomic molecules (\( \mathrm{X_2} \)).
- Intermolecular forces = London dispersion forces.
- Down the group:
- Number of electrons increases
- Electron cloud becomes more polarisable
- Stronger intermolecular forces
- More energy needed to separate molecules → higher m.p./b.p.
(ii) Electronegativity
Trend
- Electronegativity decreases down the group.
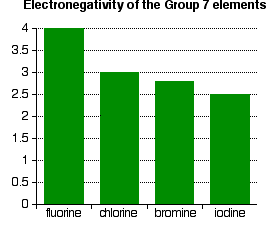
Explanation
- Atomic radius increases.
- Increased shielding effect.
- Nuclear attraction for bonding electrons decreases.
- Therefore, ability to attract electrons decreases.
Key Fact
- Fluorine is the most electronegative element.
(iii) Reactivity
Trend
- Reactivity decreases down the group.
Explanation
- Halogens react by gaining an electron:
\( \mathrm{X_2 + 2e^- \rightarrow 2X^-} \)
- Down the group:
- Atomic radius increases
- Shielding increases
- Attraction for incoming electron decreases
- Therefore, less ability to gain electron → lower reactivity
Displacement Reactions (Evidence)
- More reactive halogen displaces less reactive one:
\( \mathrm{Cl_2 + 2Br^- \rightarrow 2Cl^- + Br_2} \)
| Property | Trend Down Group | Reason |
|---|---|---|
| m.p. / b.p. | Increase | Stronger London forces |
| Electronegativity | Decrease | More shielding, larger radius |
| Reactivity | Decrease | Less attraction for electrons |
Summary
- m.p./b.p. increase due to stronger intermolecular forces.
- Electronegativity decreases due to shielding.
- Reactivity decreases due to reduced electron attraction.
Therefore, trends in Group 7 are controlled by increasing atomic size and shielding down the group.
Example 1
Explain why bromine has a higher boiling point than chlorine.
▶️ Answer/Explanation
Bromine molecules have more electrons than chlorine.
This increases polarisability of the electron cloud.
Stronger London dispersion forces between molecules.
More energy required to overcome these forces.
Therefore, bromine has a higher boiling point.
Example 2
Explain why electronegativity decreases down Group 7.
▶️ Answer/Explanation
Atomic radius increases down the group.
Increased electron shielding from inner shells.
Reduced nuclear attraction for bonding electrons.
Therefore, electronegativity decreases.
Example 3
Explain why iodine is less reactive than chlorine.
▶️ Answer/Explanation
Iodine has a larger atomic radius.
Greater shielding reduces nuclear attraction.
Less ability to gain electrons.
Therefore, iodine is less reactive.
