Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.25 Redox reactions of halogens-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.25 Redox reactions of halogens- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.25 Redox reactions of halogens- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.25 understand the trend in reactivity of Group 7 elements in terms of the redox reactions of Cl₂, Br₂ and I₂ with halide ions in aqueous solution
Students are expected to know the colours of the elements in standard conditions, in aqueous solution and in a non-polar organic solvent
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.25 Reactivity of Group 7 (Halogens) via Redox Reactions
The reactivity of halogens can be understood through redox displacement reactions with halide ions in aqueous solution.
(A) Redox
- Halogens act as oxidising agents (gain electrons).
- Halide ions act as reducing agents (lose electrons).
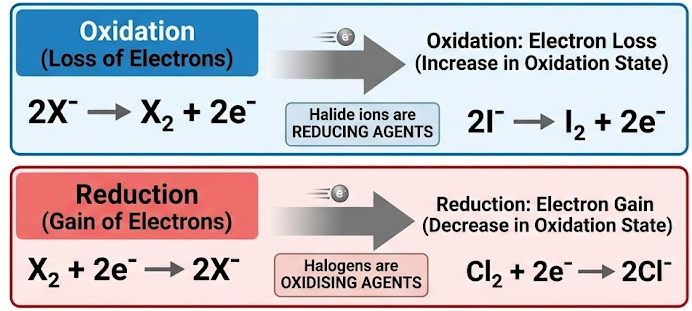
\( \mathrm{X_2 + 2e^- \rightarrow 2X^-} \)
(B) Trend in Reactivity
- Reactivity decreases down the group:
\( \mathrm{Cl_2 > Br_2 > I_2} \)
- More reactive halogen displaces less reactive halide.
(C) Displacement Reactions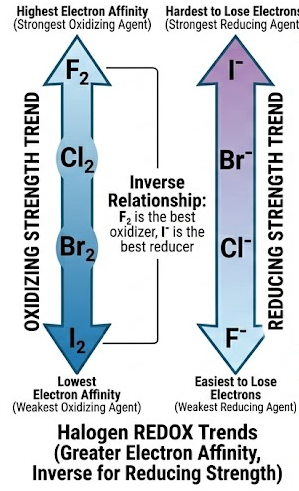
1. Chlorine + Bromide
\( \mathrm{Cl_2 + 2Br^- \rightarrow 2Cl^- + Br_2} \)
- Chlorine oxidises bromide to bromine
2. Chlorine + Iodide
\( \mathrm{Cl_2 + 2I^- \rightarrow 2Cl^- + I_2} \)
3. Bromine + Iodide
\( \mathrm{Br_2 + 2I^- \rightarrow 2Br^- + I_2} \)
No Reaction Cases
- \( \mathrm{Br_2 + Cl^-} \) → no reaction
- \( \mathrm{I_2 + Br^-} \) → no reaction
(because lower halogen is less reactive)
(D) Colours of Halogens
1. In Standard Conditions
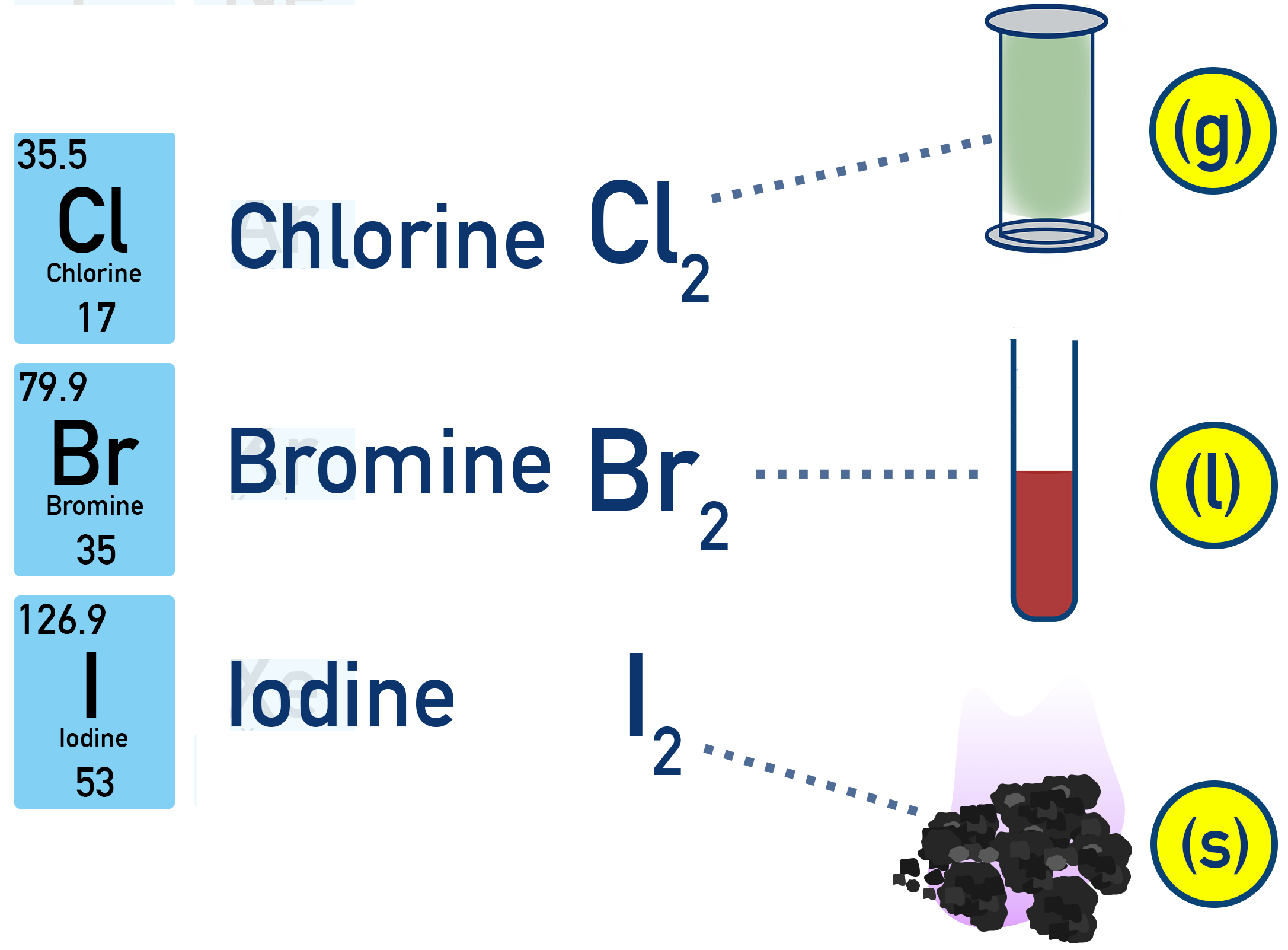
| Halogen | Colour | State |
|---|---|---|
| \( \mathrm{Cl_2} \) | Green-yellow | Gas |
| \( \mathrm{Br_2} \) | Red-brown | Liquid |
| \( \mathrm{I_2} \) | Grey solid | Solid |
2. In Aqueous Solution
| Halogen | Colour |
|---|---|
| \( \mathrm{Cl_2} \) | Pale green |
| \( \mathrm{Br_2} \) | Orange |
| \( \mathrm{I_2} \) | Brown |
3. In Non-Polar Organic Solvent (e.g. hexane)
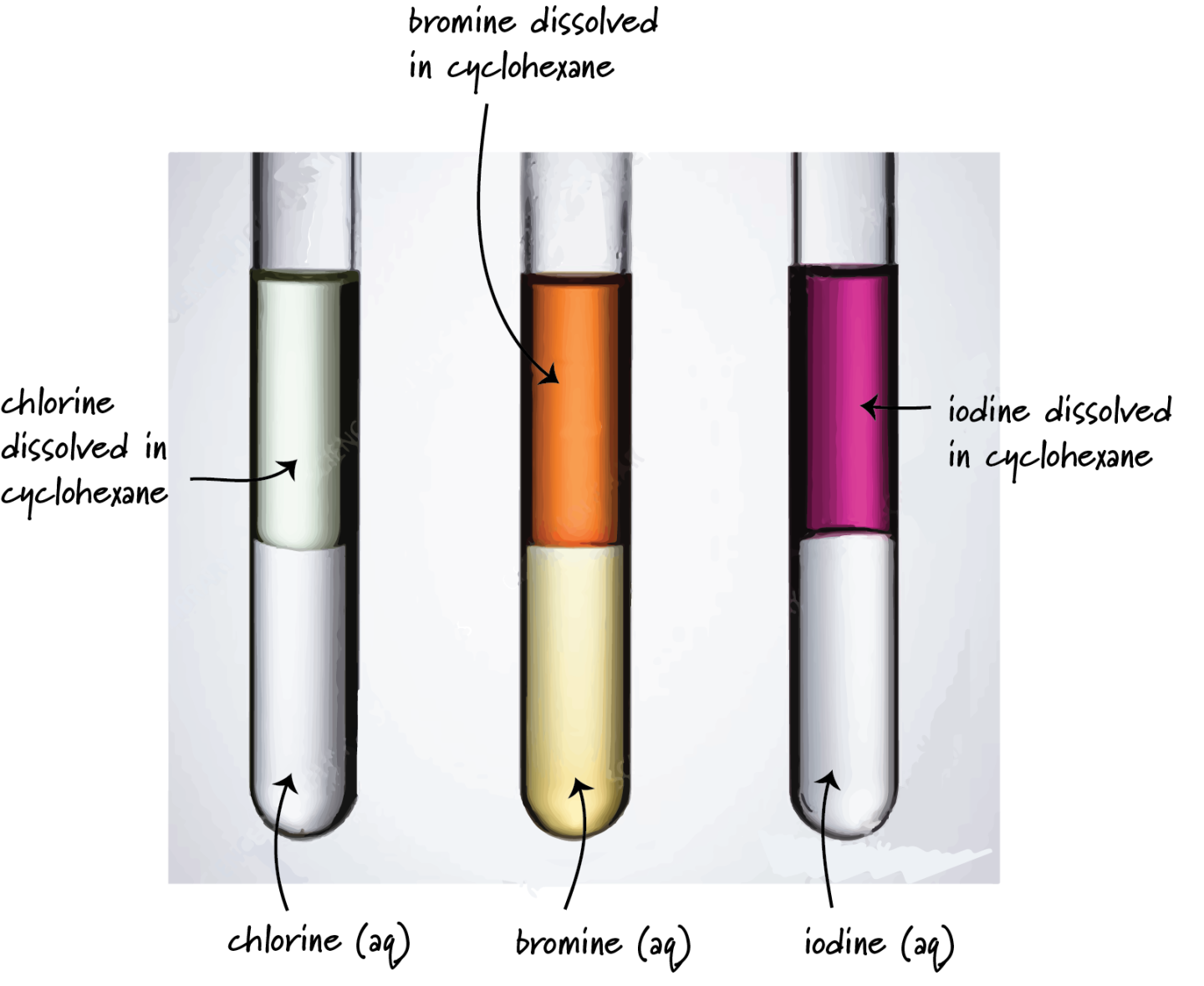
| Halogen | Colour |
|---|---|
| \( \mathrm{Cl_2} \) | Pale green |
| \( \mathrm{Br_2} \) | Orange |
| \( \mathrm{I_2} \) | Purple/violet |
(E) Explanation of Trend
- Down the group:
- Atomic radius increases
- Shielding increases
- Attraction for incoming electron decreases
- Therefore, oxidising power decreases
Summary
- Reactivity decreases down Group 7.
- More reactive halogen displaces less reactive halide.
- Colour changes help identify reactions.
Therefore, displacement reactions provide clear evidence for halogen reactivity trends.
Example 1:
Describe what you would observe when chlorine water is added to potassium iodide solution and hexane is shaken with the mixture.
▶️ Answer/Explanation
Chlorine displaces iodide to form iodine.
\( \mathrm{Cl_2 + 2I^- \rightarrow 2Cl^- + I_2} \)
Aqueous layer turns brown.
Organic (hexane) layer turns purple/violet.
Example 2
Chlorine water is added separately to aqueous potassium bromide and potassium chloride. Hexane is then added and the mixtures are shaken. Describe the observations and explain the results.
▶️ Answer/Explanation
With potassium bromide:
Chlorine displaces bromine.
\( \mathrm{Cl_2 + 2Br^- \rightarrow 2Cl^- + Br_2} \)
Aqueous layer turns orange.
Organic (hexane) layer turns orange.
With potassium chloride:
No reaction occurs.
No colour change observed.
Explanation:
- Chlorine is more reactive than bromine, so it oxidises \( \mathrm{Br^-} \) to \( \mathrm{Br_2} \).
- Chlorine cannot displace chloride ions because they are the same element.
- Therefore, reaction only occurs with less reactive halides.
