Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.26 Halogen reactions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.26 Halogen reactions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.26 Halogen reactions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.26 understand, in terms of changes in oxidation number, the following reactions of the halogens:
i oxidation reactions with Group 1 and 2 metals
ii the disproportionation reaction of chlorine with water and the use of chlorine in water treatment
iii the disproportionation reaction of chlorine with cold, dilute aqueous sodium hydroxide to form bleach
iv the disproportionation reaction of chlorine with hot alkali
v reactions analogous to those specified above
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.26 (i) Oxidation Reactions of Halogens with Group 1 and 2 Metals
Halogens react with metals to form ionic metal halides. These are redox reactions involving changes in oxidation number.
General Reactions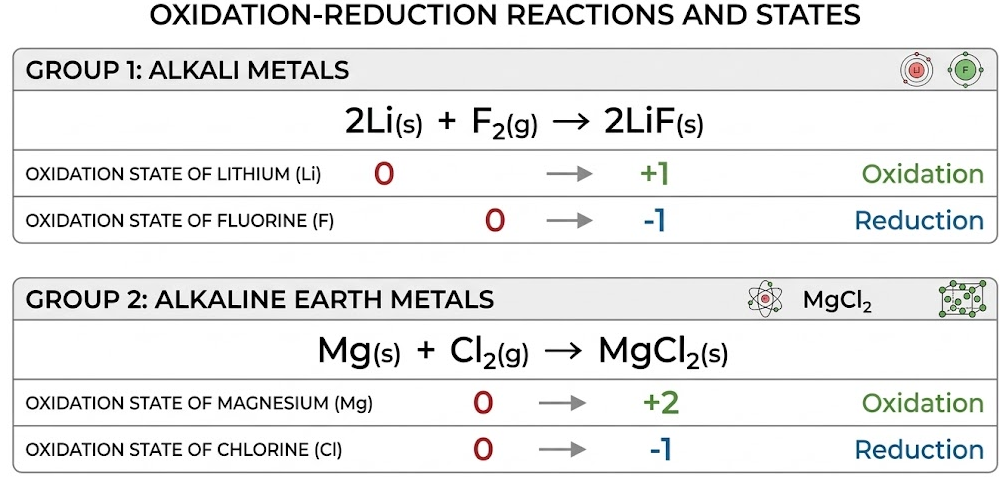
Group 1 Metals
\( \mathrm{2M + X_2 \rightarrow 2MX} \)
Group 2 Metals
\( \mathrm{M + X_2 \rightarrow MX_2} \)
Example Reactions
- \( \mathrm{2Na + Cl_2 \rightarrow 2NaCl} \)
- \( \mathrm{Mg + Cl_2 \rightarrow MgCl_2} \)
Oxidation Number Changes
Metal:
- \( \mathrm{Na: 0 \rightarrow +1} \) (oxidation)
- \( \mathrm{Mg: 0 \rightarrow +2} \) (oxidation)
Halogen:
- \( \mathrm{Cl_2: 0 \rightarrow -1} \) (reduction)
Explanation
- Metals lose electrons → oxidised
- Halogens gain electrons → reduced
- Halogens act as oxidising agents
Ionic Perspective
- \( \mathrm{Na \rightarrow Na^+ + e^-} \)
- \( \mathrm{\frac{1}{2}Cl_2 + e^- \rightarrow Cl^-} \)
Summary
- Metals are oxidised, halogens are reduced.
- Products are ionic metal halides.
- Demonstrates halogens as strong oxidising agents.
Therefore, these reactions clearly show electron transfer and oxidation number changes.
Example 1 :
Explain, in terms of oxidation numbers, the reaction between magnesium and bromine.
▶️ Answer/Explanation
\( \mathrm{Mg + Br_2 \rightarrow MgBr_2} \)
Mg: \( \mathrm{0 \rightarrow +2} \) (oxidation).
Br: \( \mathrm{0 \rightarrow -1} \) (reduction).
Therefore, Mg is oxidised and Br\(_2\) is reduced.
Example 2:
Identify the oxidising agent in the reaction \( \mathrm{2K + Cl_2 \rightarrow 2KCl} \). Justify your answer.
▶️ Answer/Explanation
Oxidising agent = \( \mathrm{Cl_2} \).
It gains electrons (reduction).
Oxidation number changes from 0 to -1.
Therefore, chlorine is the oxidising agent.
8.26 Disproportionation Reactions of Chlorine
Disproportionation is a redox reaction where the same element is both oxidised and reduced. Chlorine (\( \mathrm{Cl_2} \)) commonly undergoes disproportionation.
(ii) Chlorine with Water (Water Treatment)
Reaction
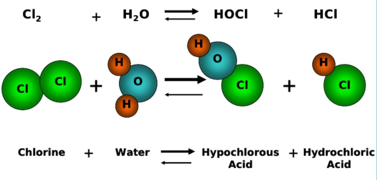
\( \mathrm{Cl_2 + H_2O \rightleftharpoons HCl + HClO} \)
Oxidation Numbers
- \( \mathrm{Cl_2: 0} \)
- In \( \mathrm{HCl} \): \( \mathrm{-1} \) → reduction
- In \( \mathrm{HClO} \): \( \mathrm{+1} \) → oxidation
Explanation
- Chlorine is both oxidised and reduced → disproportionation
- \( \mathrm{HClO} \) (hypochlorous acid) is a disinfectant
- Kills bacteria by oxidation
(iii) Chlorine with Cold, Dilute NaOH (Bleach Formation)
Reaction
\( \mathrm{Cl_2 + 2NaOH \rightarrow NaCl + NaClO + H_2O} \)
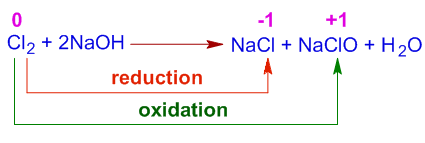
Oxidation Numbers
- \( \mathrm{Cl_2: 0} \)
- \( \mathrm{Cl^-: -1} \) → reduction
- \( \mathrm{ClO^-: +1} \) → oxidation
Explanation
- Forms sodium hypochlorite (\( \mathrm{NaClO} \))
- This is bleach (oxidising agent)
(iv) Chlorine with Hot, Concentrated NaOH
Reaction
\( \mathrm{3Cl_2 + 6NaOH \rightarrow 5NaCl + NaClO_3 + 3H_2O} \)
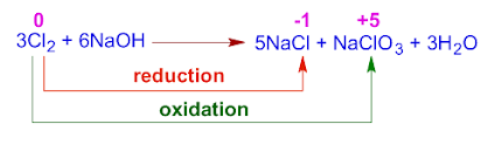 F
F
Oxidation Numbers
- \( \mathrm{Cl_2: 0} \)
- \( \mathrm{Cl^-: -1} \) → reduction
- \( \mathrm{ClO_3^-: +5} \) → oxidation
Explanation
- Stronger oxidation occurs (to +5 state).
- Forms sodium chlorate(V).
Key Comparison
| Condition | Product | Oxidation State |
|---|---|---|
| Water | \( \mathrm{HClO} \) | +1 |
| Cold dilute NaOH | \( \mathrm{ClO^-} \) | +1 |
| Hot concentrated NaOH | \( \mathrm{ClO_3^-} \) | +5 |
Summary
- Chlorine undergoes disproportionation in all cases.
- Products depend on conditions (temperature & concentration).
- Important applications: water treatment and bleach.
Therefore, oxidation state changes explain the behaviour of chlorine in different environments.
Example 3:
Explain, in terms of oxidation numbers, why the reaction of chlorine with cold NaOH is a disproportionation reaction.
▶️ Answer/Explanation
Chlorine starts at oxidation state 0.
It forms \( \mathrm{Cl^-} \) (−1) → reduction.
It also forms \( \mathrm{ClO^-} \) (+1) → oxidation.
Therefore, chlorine is both oxidised and reduced.
8.26 Analogous Reactions of Other Halogens
Other halogens (mainly bromine and iodine) undergo similar disproportionation reactions to chlorine, but are less reactive, so reactions may be slower or less complete.
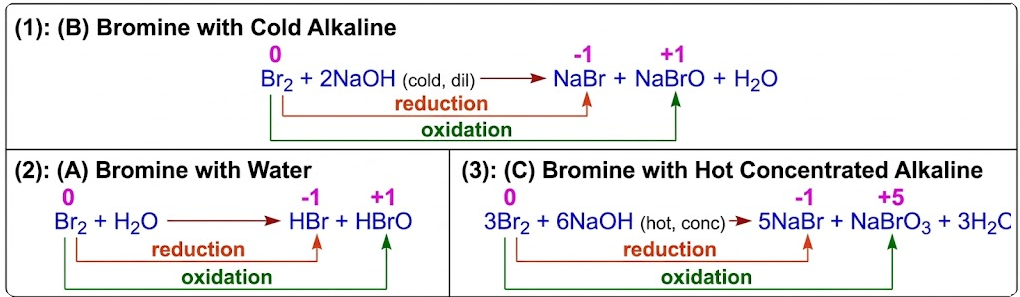
(A) Bromine with Water
\( \mathrm{Br_2 + H_2O \rightleftharpoons HBr + HBrO} \)
Oxidation Numbers
- \( \mathrm{Br_2: 0} \)
- \( \mathrm{Br^-: -1} \) → reduction
- \( \mathrm{BrO^-: +1} \) → oxidation
Note
- Similar to chlorine but less effective disinfectant.
(B) Bromine with Cold Alkaline
\( \mathrm{Br_2 + 2NaOH \rightarrow NaBr + NaBrO + H_2O} \)
- Forms bromide and hypobromite (\( \mathrm{BrO^-} \))
(C) Bromine with Hot Concentrated Alkaline
\( \mathrm{3Br_2 + 6NaOH \rightarrow 5NaBr + NaBrO_3 + 3H_2O} \)
- Forms bromide and bromate(V) (\( \mathrm{BrO_3^-} \))
(D) Iodine Reactions
With Water
- Reaction is very limited (weak disproportionation).
With Alkaline
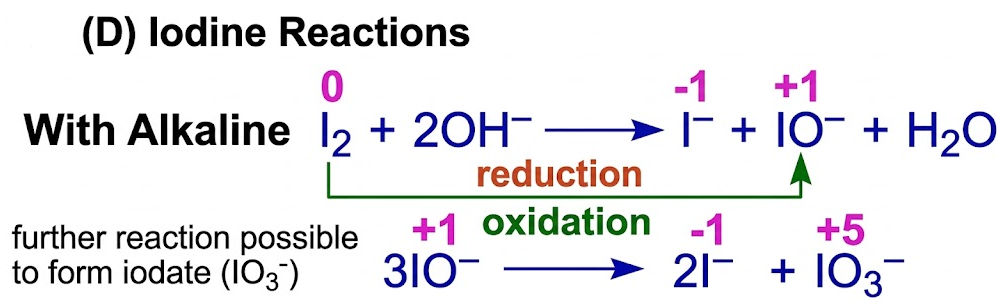
\( \mathrm{I_2 + 2OH^- \rightarrow I^- + IO^- + H_2O} \)
(further reaction possible to form iodate \( \mathrm{IO_3^-} \))
Key Trend
- Ability to disproportionate decreases down the group:
\( \mathrm{Cl_2 > Br_2 > I_2} \)
- Due to decreasing oxidising power.
Key Comparison
| Halogen | Cold Alkali | Hot Alkali |
|---|---|---|
| \( \mathrm{Cl_2} \) | \( \mathrm{Cl^- + ClO^-} \) | \( \mathrm{Cl^- + ClO_3^-} \) |
| \( \mathrm{Br_2} \) | \( \mathrm{Br^- + BrO^-} \) | \( \mathrm{Br^- + BrO_3^-} \) |
| \( \mathrm{I_2} \) | \( \mathrm{I^- + IO^-} \) | \( \mathrm{I^- + IO_3^-} \) (limited) |
Summary
- Bromine and iodine show similar disproportionation reactions to chlorine.
- Products follow same pattern: halide + oxyanion.
- Reactivity decreases down the group → reactions less extensive.
Therefore, these reactions confirm trends in oxidising ability across Group 7.
Example :
Write the equation and explain the oxidation number changes when bromine reacts with cold sodium hydroxide.
▶️ Answer/Explanation
\( \mathrm{Br_2 + 2NaOH \rightarrow NaBr + NaBrO + H_2O} \)
Br: \( \mathrm{0 \rightarrow -1} \) (reduction).
Br: \( \mathrm{0 \rightarrow +1} \) (oxidation).
Therefore, bromine is both oxidised and reduced.
