Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.27 Reactions of halides-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.27 Reactions of halides- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.27 Reactions of halides- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.27 understand the following reactions:
i solid Group 1 halides with concentrated sulfuric acid, to illustrate the trend in reducing ability of the hydrogen halides
ii precipitation reactions of the aqueous anions Cl⁻, Br⁻ and I⁻ with aqueous silver nitrate solution and nitric acid, and the solubility of the precipitates in aqueous ammonia solution
iii hydrogen halides with ammonia gas (to produce ammonium halides) and with water (to produce acids)
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.27 (i) Reaction of Solid Group 1 Halides with Concentrated Sulfuric Acid
These reactions show the trend in reducing ability of hydrogen halides (\( \mathrm{HX} \)) down Group 7.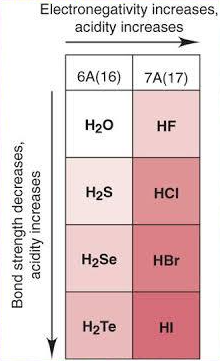
Key Trend
- Reducing ability increases down the group:
\( \mathrm{HF < HCl < HBr < HI} \)
- Due to weaker H–X bond → easier to lose electrons.
(A) Chloride (e.g. NaCl)
Reaction (Acid–Base Only)
\( \mathrm{NaCl + H_2SO_4 \rightarrow NaHSO_4 + HCl} \)
Observation

- Steamy white fumes of \( \mathrm{HCl} \)
Key Point
- No redox reaction (Cl⁻ is a weak reducing agent)
(B) Bromide (e.g. NaBr)
Step 1 (Acid–Base)
\( \mathrm{NaBr + H_2SO_4 \rightarrow NaHSO_4 + HBr} \)
Step 2 (Redox)
\( \mathrm{2HBr + H_2SO_4 \rightarrow Br_2 + SO_2 + 2H_2O} \)
Observations

- Brown fumes of \( \mathrm{Br_2} \)
- Colourless \( \mathrm{SO_2} \) gas
Key Point
- Bromide is a stronger reducing agent → reduces \( \mathrm{H_2SO_4} \)
(C) Iodide (e.g. NaI)
Step 1 (Acid–Base)
\( \mathrm{NaI + H_2SO_4 \rightarrow NaHSO_4 + HI} \)
Further Redox Reactions
- \( \mathrm{2HI + H_2SO_4 \rightarrow I_2 + SO_2 + 2H_2O} \)
- \( \mathrm{6HI + H_2SO_4 \rightarrow 3I_2 + S + 4H_2O} \)
- \( \mathrm{8HI + H_2SO_4 \rightarrow 4I_2 + H_2S + 4H_2O} \)
Observations
- Purple iodine vapour (\( \mathrm{I_2} \))
- Yellow sulfur solid (\( \mathrm{S} \))
- Rotten egg smell (\( \mathrm{H_2S} \))
Key Point
- Iodide is the strongest reducing agent
- Reduces sulfur from +6 to +4, 0, and −2
Summary Table
| Halide | Reaction Type | Products |
|---|---|---|
| \( \mathrm{Cl^-} \) | Acid–base only | \( \mathrm{HCl} \) |
| \( \mathrm{Br^-} \) | Some redox | \( \mathrm{Br_2, SO_2} \) |
| \( \mathrm{I^-} \) | Extensive redox | \( \mathrm{I_2, SO_2, S, H_2S} \) |
Summary
- Reducing ability increases down Group 7.
- Chloride → no redox, bromide → partial, iodide → extensive redox.
- Shows increasing ease of electron donation.
Therefore, reactions with sulfuric acid clearly demonstrate the trend in reducing power of halides.
Example :
Explain why iodide ions produce more products than bromide ions when reacting with concentrated sulfuric acid.
▶️ Answer/Explanation
Iodide ions are stronger reducing agents than bromide ions.
They donate electrons more readily.
Therefore, they reduce sulfuric acid further.
This produces a wider range of products (SO\(_2\), S, H\(_2\)S).
8.27 (ii) Test for Halide Ions using Silver Nitrate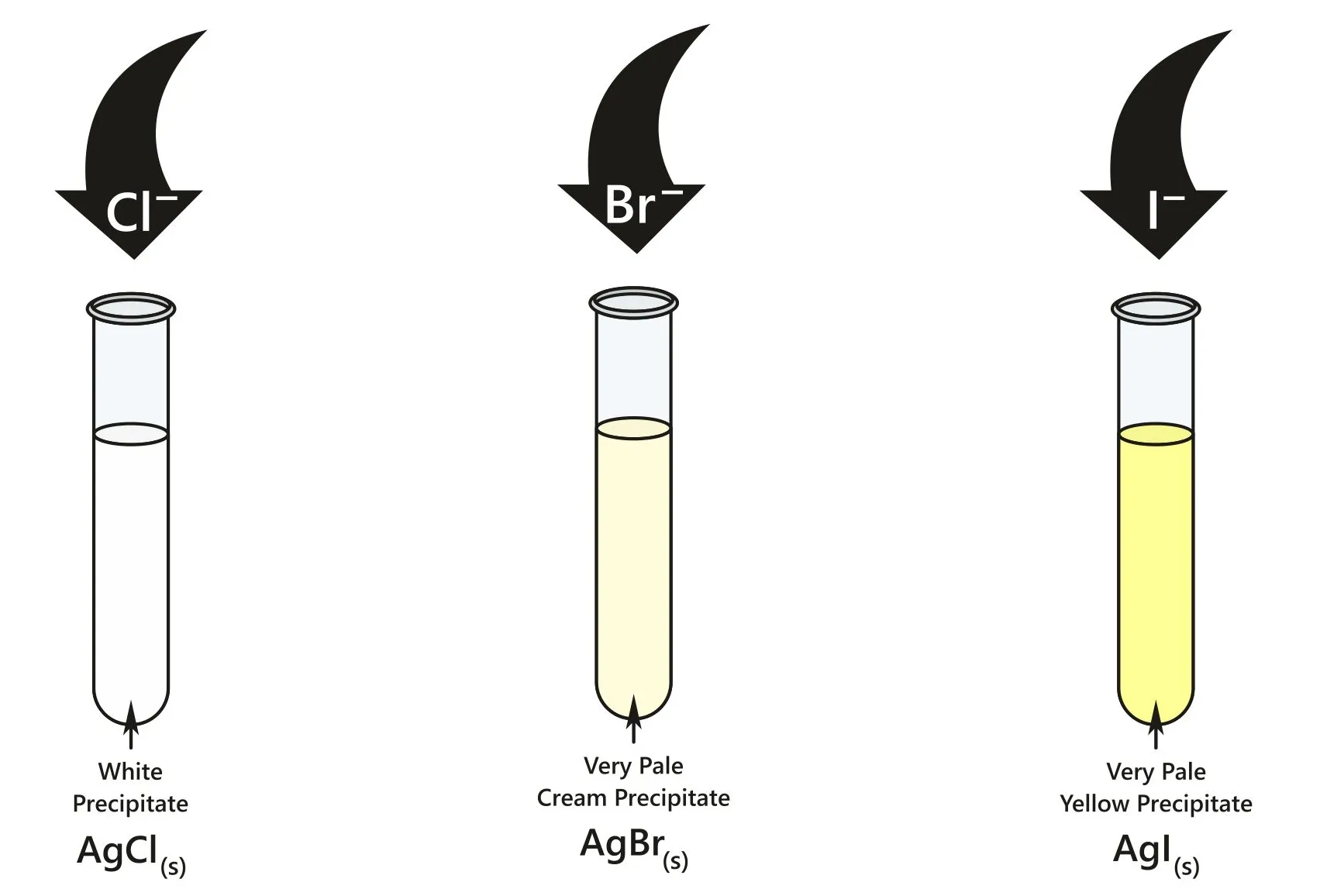
Halide ions (\( \mathrm{Cl^- , Br^- , I^-} \)) are identified using aqueous silver nitrate after acidifying with nitric acid.
(A) Why Nitric Acid is Used
- Removes interfering ions such as \( \mathrm{CO_3^{2-}} \)
- Prevents formation of other precipitates (e.g. \( \mathrm{Ag_2CO_3} \))
- Ensures test is specific for halides
(B) Precipitation Reactions
General Equation
\( \mathrm{Ag^+ + X^- \rightarrow AgX(s)} \)
Observations
| Ion | Precipitate | Colour |
|---|---|---|
| \( \mathrm{Cl^-} \) | \( \mathrm{AgCl} \) | White |
| \( \mathrm{Br^-} \) | \( \mathrm{AgBr} \) | Cream |
| \( \mathrm{I^-} \) | \( \mathrm{AgI} \) | Yellow |
(C) Solubility in Ammonia
- Used to confirm identity of halide
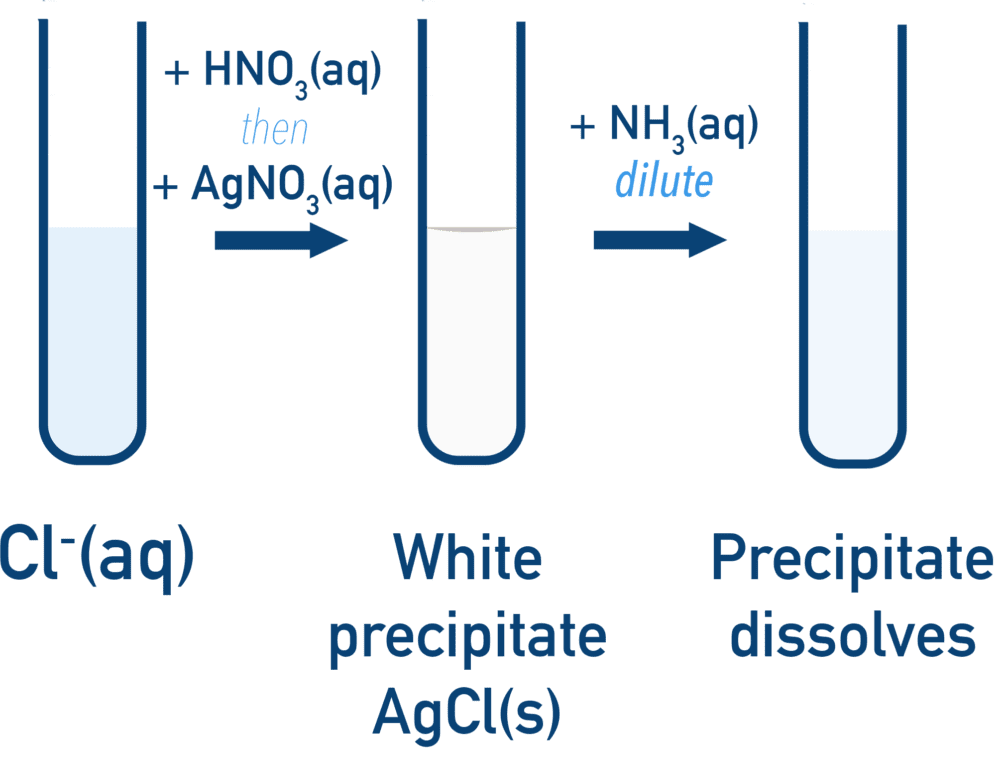
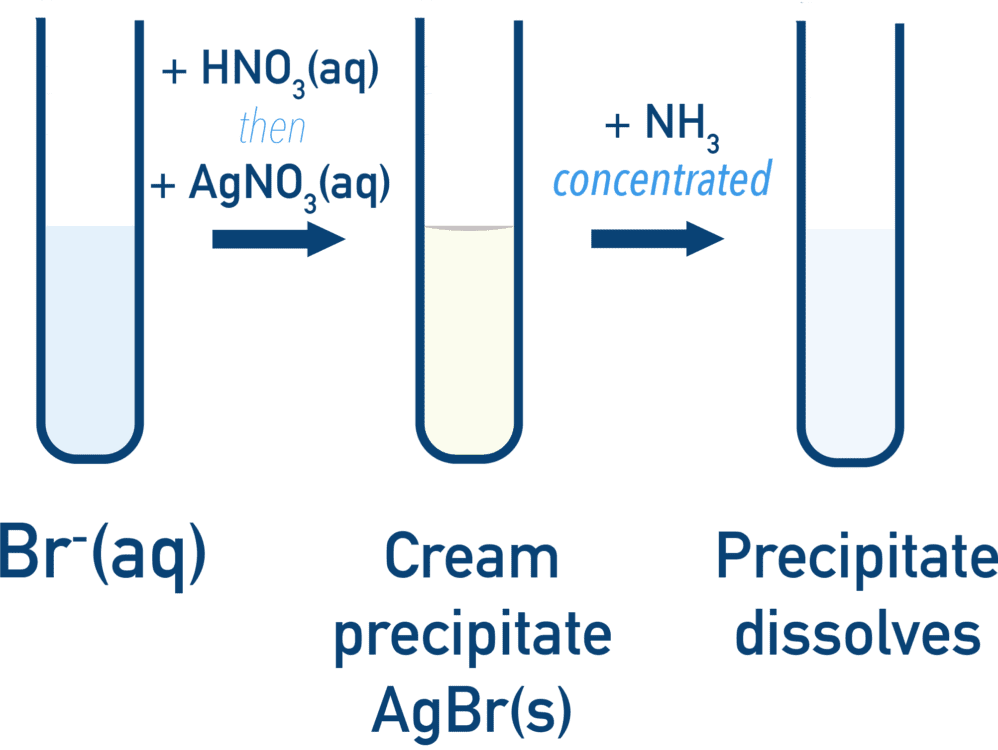
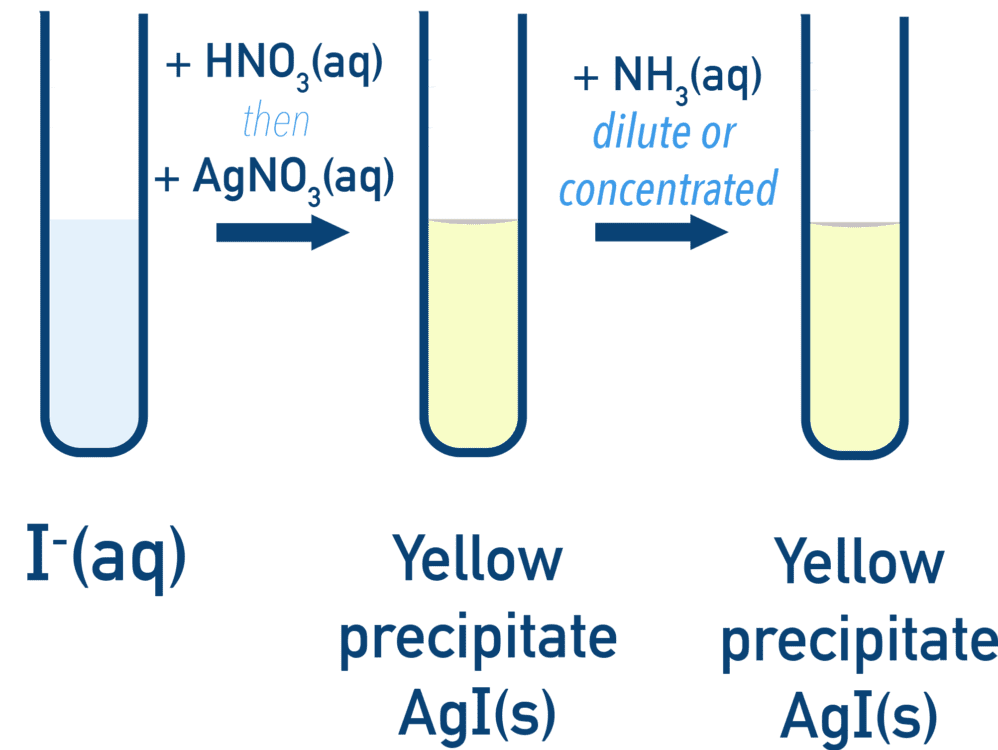
| Precipitate | Dilute NH\(_3\) | Concentrated NH\(_3\) |
|---|---|---|
| \( \mathrm{AgCl} \) | Dissolves | — |
| \( \mathrm{AgBr} \) | Partially dissolves | Dissolves |
| \( \mathrm{AgI} \) | Insoluble | Insoluble |
Explanation
- Ammonia forms a complex ion:
\( \mathrm{Ag^+ + 2NH_3 \rightarrow [Ag(NH_3)_2]^+} \)
- Strength of Ag–X bond increases from Cl → I
- Therefore solubility decreases down the group
Summary
- Silver nitrate forms characteristic precipitates.
- Colours distinguish halides.
- Ammonia confirms identity via solubility.
Therefore, this test is a key qualitative method for identifying halide ions.
Example :
A white precipitate forms with silver nitrate and dissolves in dilute ammonia. Identify the ion and explain.
▶️ Answer/Explanation
Ion = \( \mathrm{Cl^-} \).
Forms white precipitate \( \mathrm{AgCl} \).
Dissolves in dilute ammonia due to complex formation.
Therefore, chloride ion is present.
8.27 (iii) Reactions of Hydrogen Halides with Ammonia and Water
Hydrogen halides (\( \mathrm{HX} \), where X = Cl, Br, I) show important reactions with ammonia gas and water.
(A) Reaction with Ammonia Gas
General Equation
\( \mathrm{NH_3 + HX \rightarrow NH_4X} \)
Examples
- \( \mathrm{NH_3 + HCl \rightarrow NH_4Cl} \)
- \( \mathrm{NH_3 + HBr \rightarrow NH_4Br} \)
Observation

- White fumes of ammonium halide (e.g. \( \mathrm{NH_4Cl} \))
Explanation
- Ammonia acts as a base (accepts \( \mathrm{H^+} \))
- Hydrogen halide acts as an acid
- Forms ammonium salt via proton transfer
(B) Reaction with Water (Formation of Acids)
General Equation
\( \mathrm{HX + H_2O \rightarrow H_3O^+ + X^-} \)
Examples
- \( \mathrm{HCl + H_2O \rightarrow H_3O^+ + Cl^-} \)
- \( \mathrm{HBr + H_2O \rightarrow H_3O^+ + Br^-} \)
Observation

- Solution becomes acidic (low pH)
Explanation
- Hydrogen halide donates a proton to water
- Forms hydronium ions (\( \mathrm{H_3O^+} \))
- Therefore behaves as a strong acid (HCl, HBr, HI)
Key Comparison
| Reaction | Type | Product |
|---|---|---|
| With NH\(_3\) | Acid–base | Ammonium salt |
| With H\(_2\)O | Acid dissociation | \( \mathrm{H_3O^+} \) |
Summary
- Hydrogen halides react with ammonia to form ammonium salts.
- They dissolve in water to form acidic solutions.
- Both reactions involve proton transfer.
Therefore, hydrogen halides behave as acids in both gaseous and aqueous systems.
Example :
Describe the observations and explain the reaction when ammonia gas reacts with hydrogen chloride gas.
▶️ Answer/Explanation
White fumes of \( \mathrm{NH_4Cl} \) are formed.
\( \mathrm{NH_3 + HCl \rightarrow NH_4Cl} \)
Ammonia accepts a proton (base).
Hydrogen chloride donates a proton (acid).
Therefore, an ammonium salt is formed.
