Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.28 Predictions for F and At-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.28 Predictions for F and At- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.28 Predictions for F and At- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.28 be able to make predictions about fluorine and astatine and their compounds, in terms of knowledge of trends in halogen chemistry
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.28 Predictions for Fluorine and Astatine (Based on Halogen Trends)
Properties of \( \mathrm{F_2} \) and astatine (\( \mathrm{At} \)) can be predicted using trends in Group 7 (atomic size, electronegativity, reactivity, intermolecular forces).
(A) Fluorine (Top of Group)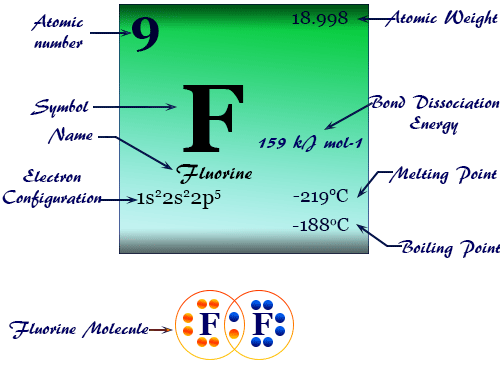
Predicted Properties
- Very low Melting Point/Boiling Point (small molecule, weak London forces)
- Gas at room temperature
- Most electronegative element
- Most reactive halogen
Explanation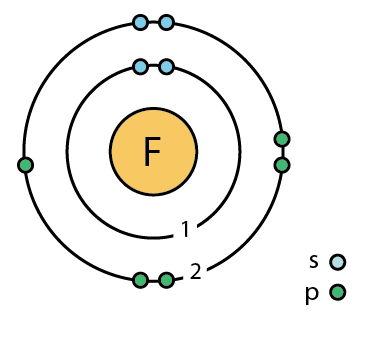
- Small atomic radius → strong attraction for electrons
- Weak F–F bond → reacts very easily
Special Behaviour (Exam Point)
- Reacts violently with water:
\( \mathrm{2F_2 + 2H_2O \rightarrow 4HF + O_2} \)
- Does not follow all trends perfectly (due to very small size)
(B) Astatine (Bottom of Group)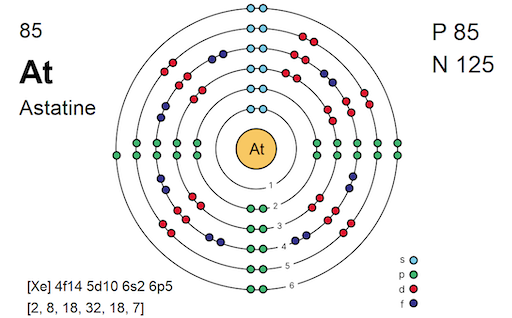
Predicted Properties
- Highest Melting Point/Boiling Point (strong London forces)
- Solid at room temperature
- Lowest electronegativity in Group 7
- Least reactive halogen
Explanation
- Very large atomic radius
- Strong shielding effect
- Weak attraction for electrons
(C) Predictions for Compounds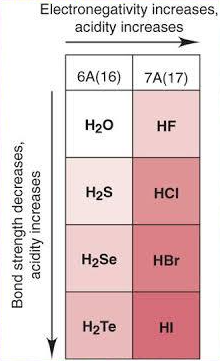
Hydrogen halides:
\( \mathrm{HF < HCl < HBr < HI < HAt} \) (increasing reducing ability)
- HF is weak acid (strong H–F bond)
- HAt expected to be very strong acid
Displacement reactions:
- Fluorine displaces all halides:
\( \mathrm{F_2 + 2Cl^- \rightarrow 2F^- + Cl_2} \)
- Astatine cannot displace any halide
| Property | Fluorine | Astatine |
|---|---|---|
| State | Gas | Solid |
| Reactivity | Highest | Lowest |
| Electronegativity | Highest | Lowest |
| Reducing ability of HX | Lowest (HF) | Highest (HAt) |
Summary
- Fluorine is extremely reactive and unusual due to small size.
- Astatine follows trends but is least reactive.
- Predictions are based on atomic size, shielding, and bond strength.
Therefore, periodic trends allow prediction of properties even for rarely studied elements like astatine.
Example :
Predict and explain the reactivity of astatine compared to iodine.
▶️ Answer/Explanation
Astatine is less reactive than iodine.
It has a larger atomic radius.
Greater electron shielding reduces nuclear attraction.
Therefore, it gains electrons less easily.
Example
Predict and explain the relative acid strength of hydrogen fluoride and hydrogen iodide in aqueous solution.
▶️ Answer/Explanation
Hydrogen iodide is a stronger acid than hydrogen fluoride.
The H–I bond is weaker than the H–F bond.
Less energy is required to break the H–I bond.
Therefore, HI dissociates more completely in water.
HF has a strong H–F bond, so it dissociates less.
Hence, HI is a stronger acid than HF.
