Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.3–8.4 Oxidation states and formula writing-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.3–8.4 Oxidation states and formula writing- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.3–8.4 Oxidation states and formula writing- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.3 be able to indicate the oxidation number of an element in a compound or an ion, using a Roman numeral
8.4 be able to write formulae given oxidation numbers
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.3 Indicating Oxidation Number using Roman Numerals
Oxidation numbers can be shown in the name of a compound using Roman numerals. This is especially important for elements that can have more than one oxidation state.
The Roman numeral indicates the oxidation number of the element in that compound.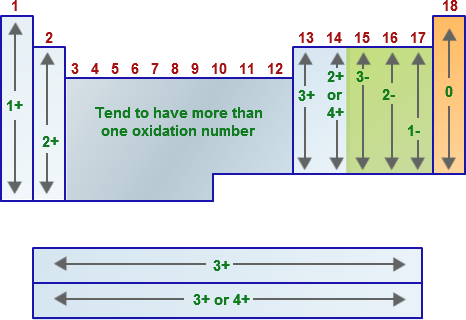
Where It Is Used
- Mainly for transition metals.
- Also for some non-metals with variable oxidation states.
Format
Name of element (Roman numeral) + compound name
Example: iron(II) chloride
Example 1: \( \mathrm{FeCl_2} \)
- Cl = −1 each → total = −2
- Fe = +2
Name: iron(II) chloride
Example 2: \( \mathrm{FeCl_3} \)
- Cl = −1 each → total = −3
- Fe = +3
Name: iron(III) chloride
Example 3: \( \mathrm{Cu_2O} \)
- O = −2
- Total Cu = +2 → each Cu = +1
Name: copper(I) oxide
Example 4: \( \mathrm{CuO} \)
- O = −2
- Cu = +2
Name: copper(II) oxide
Example 5: \( \mathrm{MnO_2} \)
- O = −2 each → total = −4
- Mn = +4
Name: manganese(IV) oxide
Why Roman Numerals Are Needed
- Some elements have multiple oxidation states.
- Without Roman numerals, names would be ambiguous.
Example:
- Iron chloride could be \( \mathrm{FeCl_2} \) or \( \mathrm{FeCl_3} \).
Therefore, Roman numerals specify the exact oxidation state.
Key Points
- Roman numerals show oxidation number.
- Written in brackets after element name.
- Essential for transition metals.
Therefore, this system ensures clear and unambiguous naming of compounds.
Example 1 :
Determine the oxidation number of chromium in \( \mathrm{CrCl_3} \) and name the compound.
▶️ Answer/Explanation
Cl = −1 each → total = −3
Cr = +3
Name: chromium(III) chloride
Example 2 :
A compound has the formula \( \mathrm{SnO_2} \). Determine the oxidation number of tin and give the name of the compound.
▶️ Answer/Explanation
O = −2 each → total = −4
Sn = +4
Name: tin(IV) oxide
8.4 Writing Formulae from Oxidation Numbers
Writing chemical formulae using oxidation numbers involves balancing the total positive and negative charges so that the compound is electrically neutral.
The total positive oxidation numbers must equal the total negative oxidation numbers.
General Method
- Write the symbols of the elements.
- Assign oxidation numbers.
- Balance charges to make total = 0.
- Simplify to smallest whole number ratio.
Method Shortcut (Criss-Cross Method)
- The magnitude of oxidation numbers are crossed over to become subscripts.
- Then simplify if possible.
Example 1: Aluminium oxide
- Al = +3, O = −2
- Balance charges: 2 × (+3) = +6, 3 × (−2) = −6
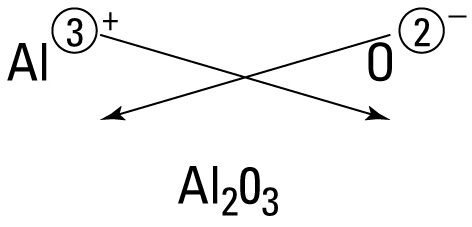
Formula: \( \mathrm{Al_2O_3} \)
Example 2: Iron(III) chloride
- Fe = +3, Cl = −1
- Need 3 Cl⁻ to balance +3

Formula: \( \mathrm{FeCl_3} \)
Example 3: Copper(I) oxide
- Cu = +1, O = −2
- Need 2 Cu⁺ to balance −2
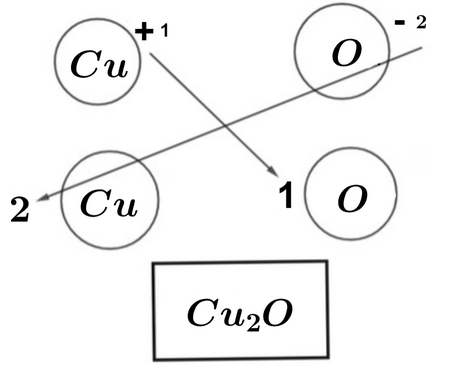
Formula: \( \mathrm{Cu_2O} \)
Example 4: Magnesium nitride
- Mg = +2, N = −3
- Balance: 3 Mg (+6) and 2 N (−6)
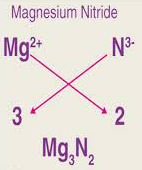
Formula: \( \mathrm{Mg_3N_2} \)
Important Notes
- Always simplify ratios to smallest whole numbers.
- Do not include charges in final formula.
- Check total charge = 0.
Key Points
- Formula is based on charge balance.
- Oxidation numbers guide subscripts.
- Criss-cross method is useful shortcut.
Therefore, writing formulae from oxidation numbers ensures correct representation of ionic compounds.
Example 1 :
Write the formula for chromium(III) oxide.
▶️ Answer/Explanation
Cr = +3, O = −2
Balance: 2 Cr (+6) and 3 O (−6)
Formula: \( \mathrm{Cr_2O_3} \)
Example 2 :
Write the formula for ammonium sulfate given \( \mathrm{NH_4^+} \) and \( \mathrm{SO_4^{2-}} \).
▶️ Answer/Explanation
NH₄⁺ = +1, SO₄²⁻ = −2
Need 2 NH₄⁺ to balance −2
Formula: \( \mathrm{(NH_4)_2SO_4} \)
