Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.5 Oxidation and reduction-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.5 Oxidation and reduction- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.5 Oxidation and reduction- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.5 understand oxidation and reduction in terms of electron transfer and changes in oxidation number, and the application of these ideas to reactions of s-block and p-block elements
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.5 Oxidation and Reduction
Oxidation and reduction (redox) reactions involve electron transfer and corresponding changes in oxidation number. These concepts are essential for understanding reactions of s-block and p-block elements.
Definitions (Electron Transfer)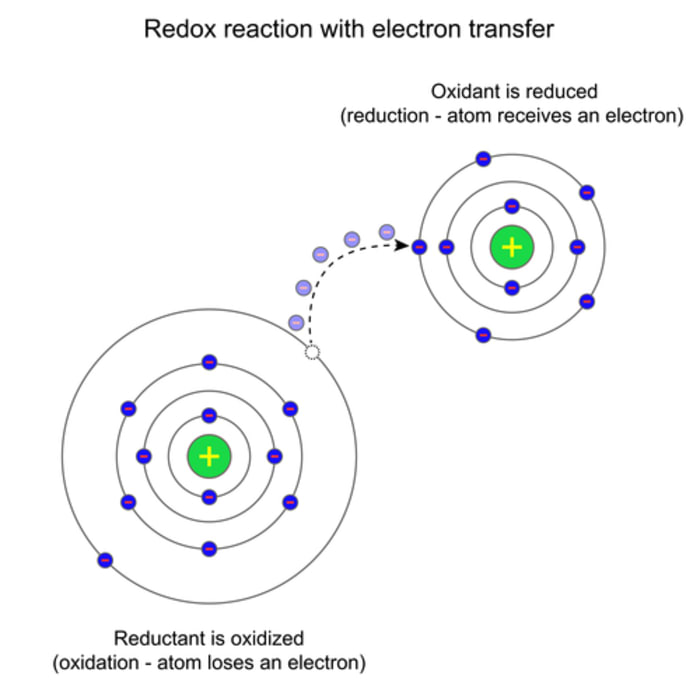
- Oxidation: loss of electrons
- Reduction: gain of electrons
Remember: OIL RIG (Oxidation Is Loss, Reduction Is Gain)
Definitions (Oxidation Number)
- Oxidation: increase in oxidation number
- Reduction: decrease in oxidation number

- Oxidation and reduction always occur together.
- Electrons lost = electrons gained.
Example 1: Reaction of Sodium with Chlorine (s-block)
\( \mathrm{2Na + Cl_2 \rightarrow 2NaCl} \)
- Na: \( \mathrm{0 \rightarrow +1} \) → oxidation (loses 1 e⁻)
- Cl: \( \mathrm{0 \rightarrow -1} \) → reduction (gains 1 e⁻)
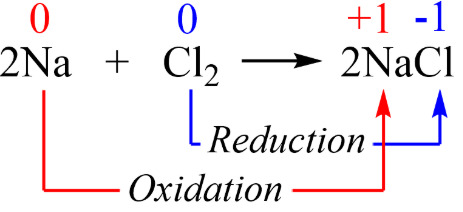
Half-equations
- \( \mathrm{Na \rightarrow Na^+ + e^-} \)
- \( \mathrm{Cl_2 + 2e^- \rightarrow 2Cl^-} \)
Therefore, sodium is oxidised and chlorine is reduced.
Example 2: Reaction of Magnesium with Oxygen (s-block)
\( \mathrm{2Mg + O_2 \rightarrow 2MgO} \)
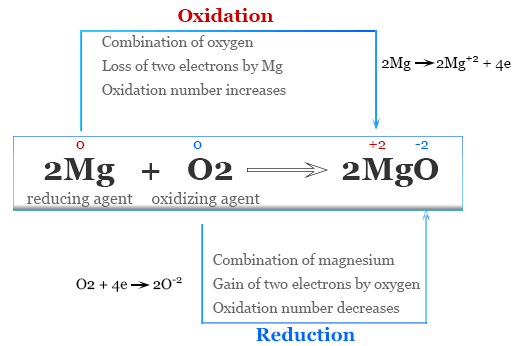
- Mg: \( \mathrm{0 \rightarrow +2} \) → oxidation
- O: \( \mathrm{0 \rightarrow -2} \) → reduction
Example 3: Reaction of Chlorine with Water (p-block)
\( \mathrm{Cl_2 + H_2O \rightarrow HCl + HClO} \)
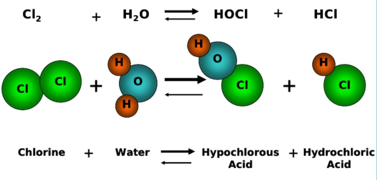
- Cl in \( \mathrm{Cl_2} \) = 0
- Cl in \( \mathrm{HCl} \) = −1 (reduction)
- Cl in \( \mathrm{HClO} \) = +1 (oxidation)
This is a disproportionation reaction (same element oxidised and reduced).
Application to s-block Elements
- s-block metals (Group 1 and 2) are easily oxidised.
- They lose electrons to form positive ions.
- Therefore, they are strong reducing agents.
Example: \( \mathrm{Na \rightarrow Na^+ + e^-} \)
Application to p-block Elements
- p-block elements show variable oxidation states.
- Can act as oxidising or reducing agents.
- Can undergo disproportionation reactions.
Key Points
- Oxidation = loss of electrons / increase in oxidation number.
- Reduction = gain of electrons / decrease in oxidation number.
- Redox reactions involve both processes simultaneously.
- s-block elements are easily oxidised.
- p-block elements show more varied redox behaviour.
Therefore, redox concepts explain the reactivity and behaviour of elements across the periodic table.
Example 1 :
Identify the oxidising and reducing agents in the reaction:
\( \mathrm{Mg + Cl_2 \rightarrow MgCl_2} \)
▶️ Answer/Explanation
Mg is oxidised (0 → +2) → reducing agent.
Cl₂ is reduced (0 → −1) → oxidising agent.
Example 2 :
Explain why chlorine can act as both an oxidising agent and a reducing agent.
▶️ Answer/Explanation
Chlorine has multiple oxidation states.
It can gain electrons (reduction) or lose electrons (oxidation).
Example: disproportionation in water.
Therefore, it can act as both oxidising and reducing agent.
