Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.6 Oxidising and reducing agents-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.6 Oxidising and reducing agents- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.6 Oxidising and reducing agents- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.6 know that oxidising agents gain electrons and reducing agents lose electrons
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.6 Oxidising Agents and Reducing Agents
In redox reactions, substances can act as oxidising agents or reducing agents depending on whether they gain or lose electrons.
Oxidising agent: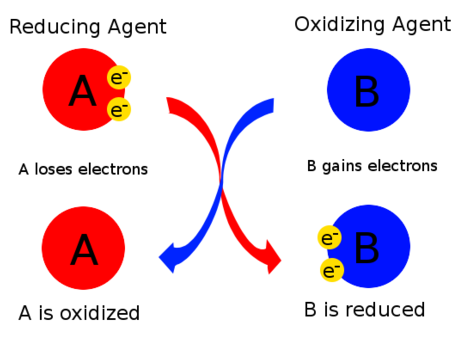
- A substance that gains electrons.
- It causes another substance to be oxidised.
Reducing agent:
- A substance that loses electrons.
- It causes another substance to be reduced.
Key Relationships
- Oxidising agent is reduced.
- Reducing agent is oxidised.
Example 1
\( \mathrm{2Na + Cl_2 \rightarrow 2NaCl} \)
- Na → \( \mathrm{Na^+ + e^-} \) (loses electrons) → reducing agent
- \( \mathrm{Cl_2 + 2e^- \rightarrow 2Cl^-} \) (gains electrons) → oxidising agent
Example 2
\( \mathrm{Mg + Cu^{2+} \rightarrow Mg^{2+} + Cu} \)
- Mg loses electrons → reducing agent
- \( \mathrm{Cu^{2+}} \) gains electrons → oxidising agent
Using Oxidation Numbers
- Oxidising agent → oxidation number decreases.
- Reducing agent → oxidation number increases.
Key Points
- Oxidising agents gain electrons.
- Reducing agents lose electrons.
- Both occur together in a redox reaction.
- Can also be identified using oxidation numbers.
Therefore, identifying oxidising and reducing agents is essential for analysing redox reactions.
Example 1:
Identify the oxidising and reducing agents in the reaction:
\( \mathrm{Zn + Fe^{2+} \rightarrow Zn^{2+} + Fe} \)
▶️ Answer/Explanation
Zn loses electrons → reducing agent.
\( \mathrm{Fe^{2+}} \) gains electrons → oxidising agent.
Example 2 :
Using oxidation numbers, identify the oxidising agent in the reaction:
\( \mathrm{Cl_2 + 2Br^- \rightarrow 2Cl^- + Br_2} \)
▶️ Answer/Explanation
Cl: \( 0 \rightarrow -1 \) (reduction)
Therefore, \( \mathrm{Cl_2} \) is the oxidising agent.
