Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.7 Disproportionation reactions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.7 Disproportionation reactions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.7 Disproportionation reactions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.7 understand that a disproportionation reaction involves an element in a single species being simultaneously oxidised and reduced
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.7 Disproportionation Reactions
A disproportionation reaction is a special type of redox reaction in which the same element in a single species is both oxidised and reduced simultaneously.
Definition
A disproportionation reaction occurs when an element undergoes both an increase and a decrease in oxidation number in the same reaction.
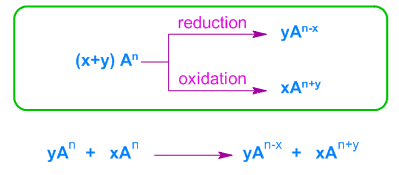
Key Idea
- One substance acts as both oxidising agent and reducing agent.
- Requires intermediate oxidation state.
Example 1: Chlorine with Water
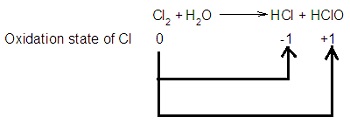
\( \mathrm{Cl_2 + H_2O \rightarrow HCl + HClO} \)
- Cl in \( \mathrm{Cl_2} \) = 0
- Cl in \( \mathrm{HCl} \) = −1 → reduction
- Cl in \( \mathrm{HClO} \) = +1 → oxidation
Therefore, chlorine is both oxidised and reduced.
Example 2: Hydrogen Peroxide Decomposition
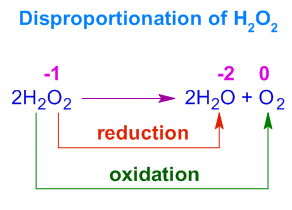
\( \mathrm{2H_2O_2 \rightarrow 2H_2O + O_2} \)
- O in \( \mathrm{H_2O_2} \) = −1
- O in \( \mathrm{H_2O} \) = −2 → reduction
- O in \( \mathrm{O_2} \) = 0 → oxidation
Example 3: Chlorine with Alkali (Cold, Dilute)
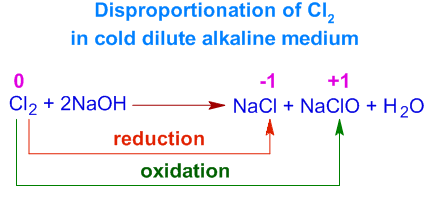
\( \mathrm{Cl_2 + 2OH^- \rightarrow Cl^- + ClO^- + H_2O} \)
- Cl: 0 → −1 (reduction)
- Cl: 0 → +1 (oxidation)
Why Disproportionation Occurs
- Element must have intermediate oxidation state.
- Can both gain and lose electrons.
Key Points
- Same element is oxidised and reduced.
- Involves two different products.
- Common in p-block elements (e.g. halogens).
Therefore, disproportionation reactions show how a single substance can act as both oxidising and reducing agent.
Example 1 :
Explain why \( \mathrm{Cl_2} \) can undergo disproportionation in water.
▶️ Answer/Explanation
Chlorine has oxidation state 0.
It can both gain electrons (to −1) and lose electrons (to +1).
Therefore, it undergoes disproportionation.
Example 2:
Identify the oxidation and reduction processes in:
\( \mathrm{2H_2O_2 \rightarrow 2H_2O + O_2} \)
▶️ Answer/Explanation
O: −1 → −2 (reduction).
O: −1 → 0 (oxidation).
Therefore, disproportionation occurs.
