Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.8 Oxidation number in classification-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.8 Oxidation number in classification- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.8 Oxidation number in classification- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.8 know that oxidation number is a useful concept in terms of the classification of reactions as redox and as disproportionation
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.8 Oxidation Number and its Use in Redox & Disproportionation
The concept of oxidation number (oxidation state) is used to track the movement of electrons in chemical reactions and to classify reactions as redox or disproportionation.
Definition of Oxidation Number
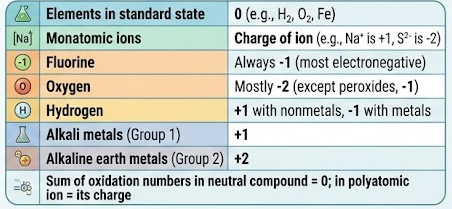
The oxidation number is the charge that an atom would have if all bonds were ionic.
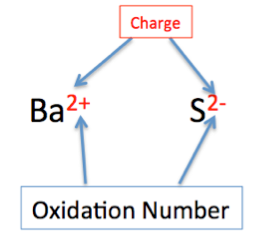
- Helps identify electron loss or gain.
- Used to analyse redox processes.
Redox Reactions
A redox reaction involves both oxidation and reduction.
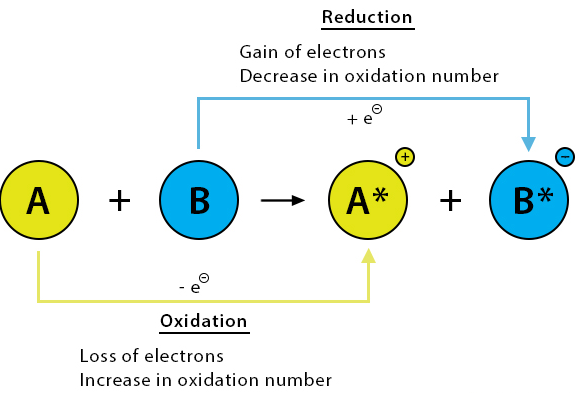
- Oxidation → increase in oxidation number (loss of electrons)
- Reduction → decrease in oxidation number (gain of electrons)
Example
\( \mathrm{Zn + Cu^{2+} \rightarrow Zn^{2+} + Cu} \)
- Zn: \( 0 \rightarrow +2 \) (oxidation)
- Cu: \( +2 \rightarrow 0 \) (reduction)
Therefore, oxidation numbers clearly show electron transfer in redox reactions.
Disproportionation Reactions
Disproportionation is a special type of redox reaction in which the same element is both oxidised and reduced.
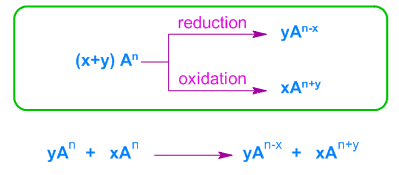
Example
\( \mathrm{Cl_2 + H_2O \rightarrow HCl + HClO} \)
- Cl in \( \mathrm{Cl_2} \): 0
- Cl in \( \mathrm{HCl} \): −1 (reduction)
- Cl in \( \mathrm{HClO} \): +1 (oxidation)
Therefore, chlorine is simultaneously oxidised and reduced.
Why Oxidation Number is Useful
- Identifies whether a reaction is redox.
- Shows which species is oxidised or reduced.
- Helps detect disproportionation reactions.
- Useful in balancing redox equations.
Key Exam Points
- Increase in oxidation number = oxidation.
- Decrease in oxidation number = reduction.
- Same element oxidised and reduced = disproportionation.
Therefore, oxidation number is a key concept for analysing and classifying chemical reactions.
Example 1 :
Determine whether the following reaction is a redox reaction and identify the species oxidised and reduced:
\( \mathrm{2Fe^{2+} + Cl_2 \rightarrow 2Fe^{3+} + 2Cl^-} \)
▶️ Answer/Explanation
Fe: \( +2 \rightarrow +3 \) (oxidation)
Cl: \( 0 \rightarrow -1 \) (reduction)
Therefore, the reaction is a redox reaction.
Example 2 :
Show that the following reaction is a disproportionation reaction using oxidation numbers:
\( \mathrm{2H_2O_2 \rightarrow 2H_2O + O_2} \)
▶️ Answer/Explanation
O in \( \mathrm{H_2O_2} \): −1
O in \( \mathrm{H_2O} \): −2 (reduction)
O in \( \mathrm{O_2} \): 0 (oxidation)
The same element (oxygen) is both oxidised and reduced.
Therefore, this is a disproportionation reaction.
