Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.9 Formation of ions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.9 Formation of ions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.9 Formation of ions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.9 understand that metals, in general, form positive ions by loss of electrons with an increase in oxidation number whereas non-metals, in general, form negative ions by gain of electrons with a decrease in oxidation number
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.9 Formation of Ions and Changes in Oxidation Number
The behaviour of elements in forming ions can be understood using the concept of oxidation number and electron transfer.
Metals: Formation of Positive Ions (Cations)
Metals tend to lose electrons to form positive ions (cations).
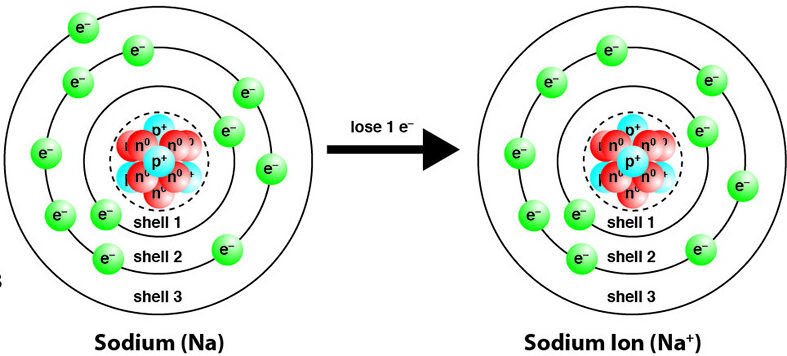
- Loss of electrons = oxidation.
- Oxidation number increases.
Example
\( \mathrm{Na \rightarrow Na^+ + e^-} \)
- Na: \( 0 \rightarrow +1 \)
- Oxidation number increases.
Therefore, metals are electron donors and are oxidised.
Non-metals: Formation of Negative Ions (Anions)
Non-metals tend to gain electrons to form negative ions (anions).
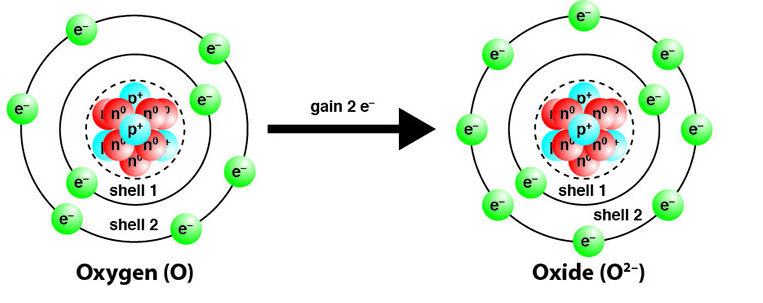
- Gain of electrons = reduction.
- Oxidation number decreases.
Example
\( \mathrm{Cl + e^- \rightarrow Cl^-} \)
- Cl: \( 0 \rightarrow -1 \)
- Oxidation number decreases.
Therefore, non-metals are electron acceptors and are reduced.
Summary of Trends
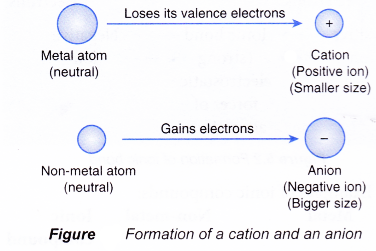
- Metals → lose electrons → form cations → oxidation number increases.
- Non-metals → gain electrons → form anions → oxidation number decreases.
Link to Redox Reactions
- Metals are usually oxidised (reducing agents).
- Non-metals are usually reduced (oxidising agents).
Example Reaction
\( \mathrm{2Na + Cl_2 \rightarrow 2NaCl} \)
- Na: \( 0 \rightarrow +1 \) (oxidation)
- Cl: \( 0 \rightarrow -1 \) (reduction)
Exam Points
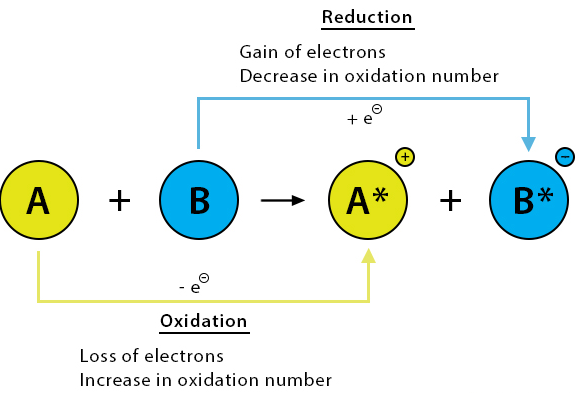
- Loss of electrons → oxidation → increase in oxidation number.
- Gain of electrons → reduction → decrease in oxidation number.
- Metals form positive ions; non-metals form negative ions.
Therefore, oxidation number provides a clear way to understand how metals and non-metals behave in reactions.
Example 1 :
In the reaction:
\( \mathrm{2Al + 3Cl_2 \rightarrow 2AlCl_3} \)
(a) Determine the oxidation number changes for aluminium and chlorine.
(b) Explain, in terms of electron transfer, why aluminium acts as a reducing agent.
▶️ Answer/Explanation
(a)
Al: \( 0 \rightarrow +3 \)
Cl: \( 0 \rightarrow -1 \)
(b)
Aluminium loses three electrons per atom.
This increases its oxidation number → oxidation.
It supplies electrons to chlorine, causing chlorine to be reduced.
Therefore, aluminium acts as a reducing agent.
Example 2 :
A student states: “All non-metals act as oxidising agents.”
Using oxidation numbers and a suitable reaction, evaluate this statement.
▶️ Answer/Explanation
The statement is not always true.
Example:
\( \mathrm{Cl_2 + H_2O \rightarrow HCl + HClO} \)
Cl: \( 0 \rightarrow -1 \) (reduction)
Cl: \( 0 \rightarrow +1 \) (oxidation)
Chlorine is both reduced and oxidised → disproportionation.
Therefore, non-metals can act as both oxidising and reducing agents depending on the reaction.
