Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.1 Collision theory-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.1 Collision theory- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.1 Collision theory- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.1 understand, in terms of the collision theory, the effect of changes in concentration, temperature, pressure and surface area on the rate of a chemical reaction
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.1 Collision Theory and Factors Affecting Rate of Reaction
The rate of a chemical reaction depends on how frequently reacting particles collide and whether these collisions are successful.
Collision Theory
Collision theory states that for a reaction to occur, particles must collide with sufficient energy and correct orientation.
![]()
- Not all collisions lead to a reaction.
- Only collisions with energy greater than or equal to the activation energy are successful.
- The rate of reaction depends on the number of successful collisions per unit time.
Therefore, any factor that increases the frequency of collisions or the number of successful collisions will increase the rate of reaction.
Effect of Concentration
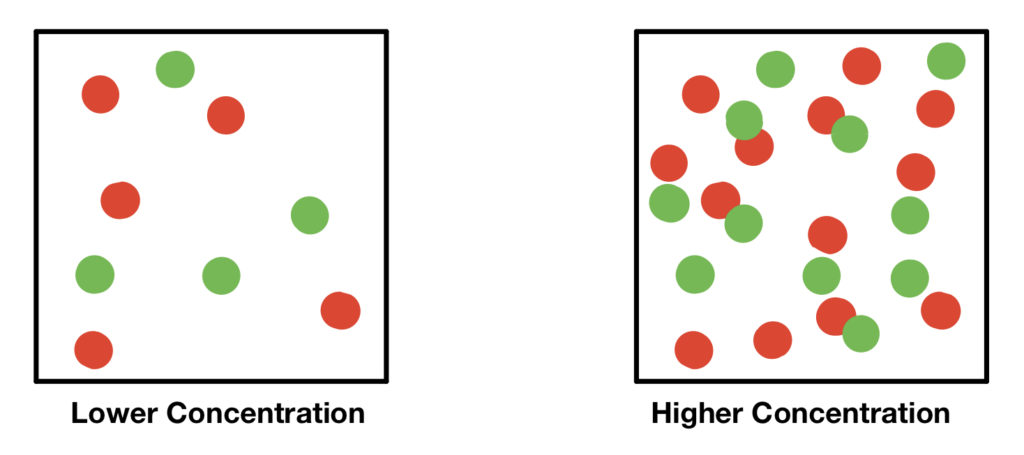
- Increasing the concentration of reactants increases the number of particles per unit volume.
- This leads to more frequent collisions between particles.
- As a result, the number of successful collisions per unit time increases.
- Therefore, the rate of reaction increases.
Decreasing concentration has the opposite effect, reducing collision frequency and lowering the rate.
Effect of Temperature
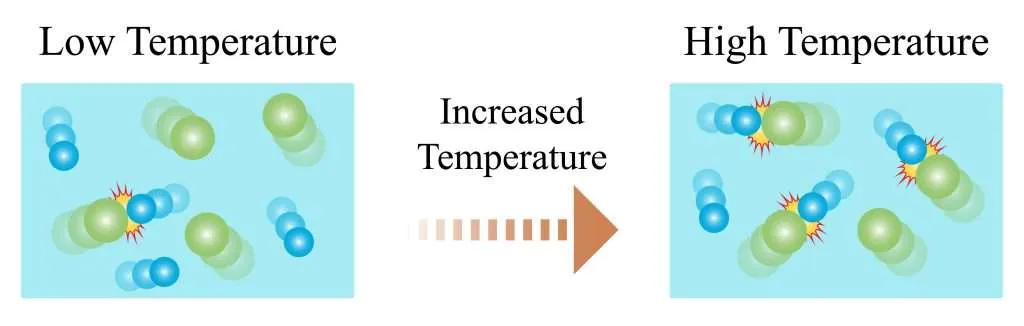
- Increasing temperature increases the kinetic energy of particles.
- Particles move faster, leading to more frequent collisions.
- A greater proportion of particles have energy equal to or greater than the activation energy.
- This significantly increases the number of successful collisions.
- Therefore, the rate of reaction increases.
Temperature has a greater effect than concentration because it increases both collision frequency and the fraction of successful collisions.
Effect of Pressure (for gases)
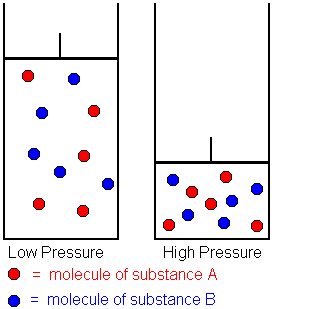
- Increasing pressure decreases the volume of the gas.
- Gas particles become closer together.
- This increases the frequency of collisions.
- As a result, the number of successful collisions per unit time increases.
- Therefore, the rate of reaction increases.
Decreasing pressure increases volume, reducing collision frequency and lowering the rate.
Effect of Surface Area
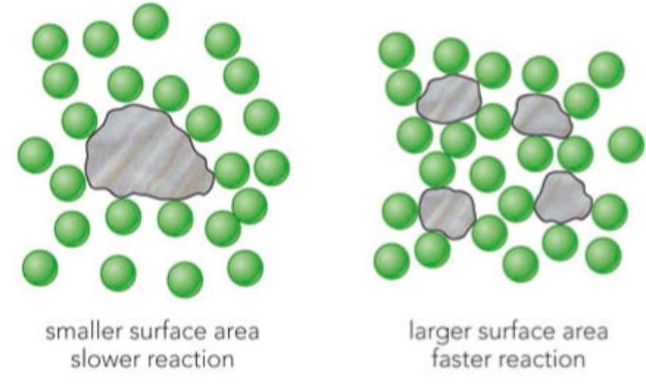
- Increasing surface area (e.g. by powdering a solid) exposes more particles to react.
- More particles are available for collisions at the surface.
- This increases the frequency of collisions between reactants.
- As a result, the number of successful collisions increases.
- Therefore, the rate of reaction increases.
Larger solid pieces have a smaller surface area, resulting in fewer collisions and a slower reaction rate.
Example 1:
Explain why increasing temperature increases the rate of a reaction.
▶️ Answer/Explanation
Increasing temperature increases the kinetic energy of particles.
This causes particles to move faster and collide more frequently.
A greater proportion of particles have energy greater than or equal to the activation energy.
Therefore, the number of successful collisions increases, increasing the rate of reaction.
Example 2:
Explain why powdered calcium carbonate reacts faster with hydrochloric acid than large lumps.
▶️ Answer/Explanation
Powdered calcium carbonate has a larger surface area than large lumps.
More particles are exposed and available for collisions.
This increases the frequency of collisions with hydrochloric acid particles.
Therefore, the number of successful collisions increases, leading to a faster reaction rate.
