Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.10 Effect of conditions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.10 Effect of conditions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.10 Effect of conditions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.10 be able to predict and justify the qualitative effects of changes of temperature, pressure and concentration on the position of equilibrium in a homogeneous system
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.10 Effects of Temperature, Pressure and Concentration on the Position of Equilibrium
The position of equilibrium in a homogeneous system can be altered by changing external conditions such as temperature, pressure and concentration.

These changes affect the rates of the forward and backward reactions differently, causing the equilibrium to shift in a direction that opposes the change. This behaviour is explained by Le Chatelier’s Principle.
Le Chatelier’s Principle
When a system at equilibrium is subjected to a change in conditions, the equilibrium shifts in the direction that opposes the change.
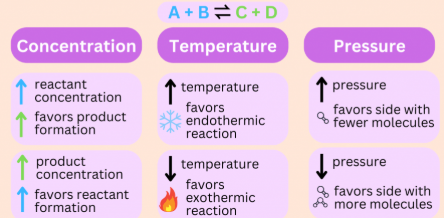
Effect of Concentration
- Increasing the concentration of a reactant shifts the equilibrium towards the products (forward reaction) to reduce the added reactant.
- Increasing the concentration of a product shifts the equilibrium towards the reactants (backward reaction).
- Decreasing the concentration of a species shifts the equilibrium in the direction that produces more of that species.

This occurs because the rate of the reaction involving the added substance increases immediately, disturbing equilibrium and causing a shift until a new equilibrium is established.
Effect of Pressure (Gaseous Systems Only)
- Increasing pressure shifts the equilibrium towards the side with fewer moles of gas.
- Decreasing pressure shifts the equilibrium towards the side with more moles of gas.
- If both sides have equal numbers of gas molecules, there is no change in equilibrium position.

This happens because increasing pressure effectively increases gas concentration, so the system shifts to reduce pressure by favouring the side with fewer gas particles.
Effect of Temperature
- Increasing temperature shifts equilibrium in the endothermic direction.
- Decreasing temperature shifts equilibrium in the exothermic direction.
This can be understood by treating heat as a reactant or product:
- In an exothermic reaction, heat is released, so increasing temperature shifts equilibrium backwards.
- In an endothermic reaction, heat is absorbed, so increasing temperature shifts equilibrium forwards.

Temperature is the only factor that changes the value of the equilibrium constant because it affects the relative energies of reactants and products.
Illustration with a Reversible Reaction
\( \mathrm{N_2(g) + 3H_2(g) \ \rightleftharpoons \ 2NH_3(g)} \ \ \Delta H < 0 \)
- Increasing pressure shifts equilibrium to the right (fewer gas moles).
- Increasing temperature shifts equilibrium to the left (endothermic direction).
- Increasing \( \mathrm{H_2} \) concentration shifts equilibrium to the right.
Example 1:
Explain how increasing pressure affects the position of equilibrium in the Haber process.
▶️ Answer/Explanation
In the Haber process, \( \mathrm{N_2(g) + 3H_2(g) \ \rightleftharpoons \ 2NH_3(g)} \), there are 4 moles of gas on the left and 2 moles on the right.
Increasing pressure shifts the equilibrium towards the side with fewer gas molecules to reduce the pressure.
Therefore, the equilibrium shifts to the right, increasing the yield of \( \mathrm{NH_3} \).
Example 2:
The forward reaction in a system is exothermic. Predict and explain the effect of increasing temperature on the equilibrium position.
▶️ Answer/Explanation
In an exothermic reaction, heat is released in the forward direction.
Increasing temperature adds heat to the system, so the equilibrium shifts in the endothermic direction to oppose this change.
Therefore, the equilibrium shifts towards the reactants (backward reaction).
This results in a decrease in the yield of products.
