Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.11 Industrial compromise-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.11 Industrial compromise- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.11 Industrial compromise- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.11 evaluate data to explain the necessity, for many industrial processes, to reach a compromise between the yield and the rate of reaction
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.11 Compromise between Yield and Rate in Industrial Processes
In many industrial processes, reversible reactions are used to produce desired products. However, the conditions that give the highest equilibrium yield are often different from those that give a fast rate of reaction. As a result, industrial chemists must choose operating conditions that provide a compromise between achieving a high yield and maintaining a sufficiently fast reaction rate.
Nature of the Compromise
Industrial conditions are selected to balance economic efficiency by achieving an acceptable yield within a reasonable time.
The position of equilibrium determines the maximum possible yield, while the rate of reaction determines how quickly that yield is reached. In practice, operating at conditions that favour one often disadvantages the other.
Effect of Temperature
- Increasing temperature increases the rate of reaction because particles have more kinetic energy and collide more frequently with sufficient energy.
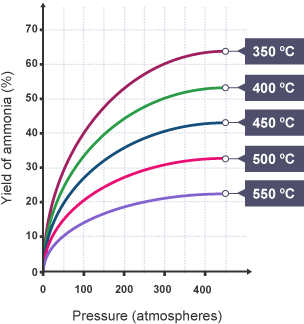
- However, for exothermic reactions, increasing temperature shifts equilibrium towards the reactants, reducing yield.
- Lower temperatures favour higher yield but result in a very slow reaction rate.
Therefore, a moderate temperature is chosen to balance a reasonable rate with an acceptable yield.
Effect of Pressure (for Gaseous Systems)
- Increasing pressure increases the rate of reaction by increasing the frequency of collisions between gas particles.
- It also shifts equilibrium towards the side with fewer moles of gas, increasing yield if this favours products.
- However, very high pressures are expensive and require strong equipment, increasing costs and safety risks.
Therefore, a compromise pressure is used that improves yield and rate without excessive cost.
Illustration: Haber Process
\( \mathrm{N_2(g) + 3H_2(g) \ \rightleftharpoons \ 2NH_3(g)} \ \ \Delta H < 0 \)
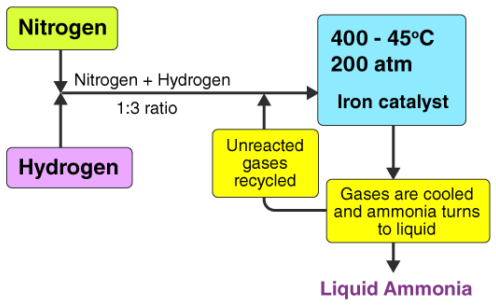
- Low temperature favours high yield of \( \mathrm{NH_3} \), but the reaction is too slow.
- High temperature increases rate but reduces yield.
- High pressure increases both rate and yield, but is costly.
- Therefore, conditions such as moderate temperature, high (but not extreme) pressure, and an iron catalyst are used.
Role of Catalysts
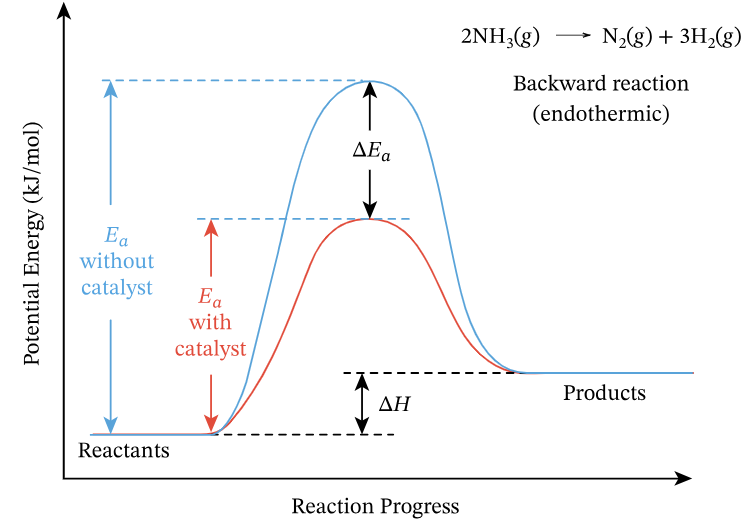
- Catalysts increase the rate of both forward and backward reactions by providing an alternative pathway with lower activation energy.
- They do not change the position of equilibrium or the equilibrium yield.
- Catalysts allow equilibrium to be reached more quickly under compromise conditions.
Key Idea
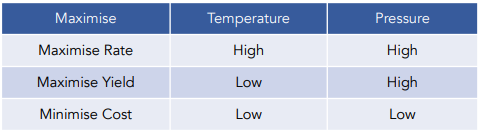
- Industrial processes prioritise overall efficiency, not just maximum yield.
- A slightly lower yield may be accepted if the reaction occurs significantly faster.
- Recycling of unreacted reactants is often used to improve overall yield.
Example 1:
Explain why a high temperature is not used in the Haber process despite increasing the rate of reaction.
▶️ Answer/Explanation
Although a high temperature increases the rate of reaction by providing particles with more kinetic energy, the forward reaction in the Haber process is exothermic.
Increasing temperature shifts the equilibrium towards the endothermic direction, which is the backward reaction.
This reduces the equilibrium yield of \( \mathrm{NH_3} \).
Therefore, a compromise temperature is used to balance rate and yield.
Example 2:
Explain why extremely high pressure is not always used in industrial equilibrium processes.
▶️ Answer/Explanation
Increasing pressure can increase both the rate of reaction and the yield if the equilibrium favours fewer gas molecules.
However, extremely high pressures require strong, expensive equipment and increase energy costs.
There are also safety risks associated with operating at very high pressures.
Therefore, a compromise pressure is chosen to balance economic cost, safety, and efficiency.
