Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.2 Activation energy-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.2 Activation energy- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.2 Activation energy- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.2 understand that reactions take place only when collisions have sufficient energy, known as the activation energy
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.2 Activation Energy and Successful Collisions
For a chemical reaction to occur, colliding particles must have sufficient energy. This minimum energy requirement is explained by the concept of activation energy.
Activation Energy
Activation energy is the minimum energy that colliding particles must possess for a reaction to occur.
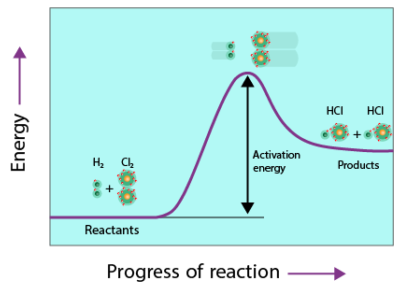
This energy is required to break existing bonds in the reactants before new bonds can form to produce products.
If particles collide with energy less than the activation energy, the collision will not result in a reaction.
Successful and Unsuccessful Collisions
Collisions between particles can be classified based on whether they lead to a reaction.
Successful Collisions

- Occur when particles collide with energy greater than or equal to the activation energy.
- Particles must also have the correct orientation for bonds to break and form.
- These collisions lead to the formation of products.
Therefore, only successful collisions result in a chemical reaction.
Unsuccessful Collisions
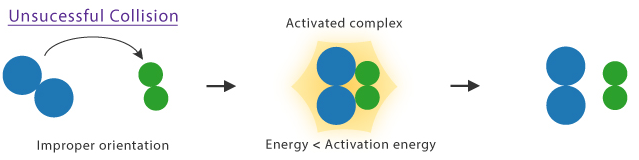
- Occur when particles collide with energy less than the activation energy.
- Collisions may also be unsuccessful if particles are not correctly oriented.
- In these cases, no reaction occurs and particles simply separate.
As a result, many collisions in a reaction mixture are unsuccessful, and only a fraction lead to product formation.
Energy Profile Explanation
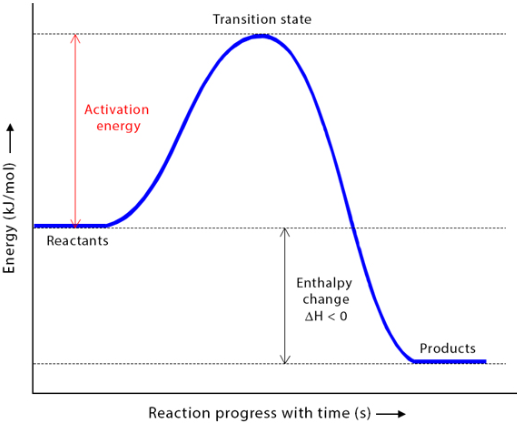
- During a reaction, energy must be supplied to reach the activation energy barrier.
- Once this barrier is overcome, bonds can break and new bonds can form.
- The activation energy represents the energy difference between reactants and the transition state.
Therefore, reactions only occur when particles collide with sufficient energy to overcome this barrier.
Example 1:
Explain why not all collisions result in a reaction.
▶️ Answer/Explanation
Not all collisions have sufficient energy to overcome the activation energy.
Some collisions also occur with incorrect orientation.
Therefore, these collisions are unsuccessful and do not result in a reaction.
Example 2:
Explain the role of activation energy in a chemical reaction.
▶️ Answer/Explanation
Activation energy is the minimum energy required for a reaction to occur.
It is needed to break existing bonds in the reactants.
Only collisions with energy greater than or equal to this value are successful.
Therefore, activation energy determines whether a reaction will occur.
