Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.3 Rate calculations-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.3 Rate calculations- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.3 Rate calculations- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.3 be able to calculate the rate of a reaction from:
i the time taken for a reaction, using rate = 1/time
ii the gradient of suitable graph, by drawing a tangent, either for initial rate, or at a time, t
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.3(i) Calculating Rate of Reaction using Time
The rate of a reaction can be determined by measuring how long a reaction takes to complete.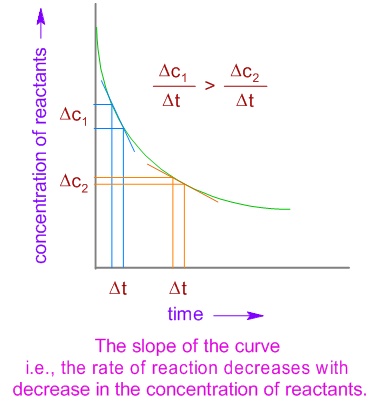
Rate from Time
The rate of reaction is inversely proportional to the time taken.
rate = \( \frac{1}{\text{time}} \)
- A shorter time means a faster reaction.
- A longer time means a slower reaction.
This method is commonly used when measuring the time taken for a visible change, such as:
- Formation of a precipitate (e.g. solution becoming cloudy).
- Disappearance of a solid.
- Change in colour.
Since it may be difficult to measure the exact rate directly, using \( \frac{1}{\text{time}} \) provides a convenient way to compare reaction rates.
Interpretation
- If the time taken is small, \( \frac{1}{\text{time}} \) is large → reaction is fast.
- If the time taken is large, \( \frac{1}{\text{time}} \) is small → reaction is slow.
Therefore, the value of \( \frac{1}{\text{time}} \) gives a measure of how quickly the reaction occurs.
Example 1:
A reaction takes 50 seconds to complete. Calculate the rate of reaction.
▶️ Answer/Explanation
rate = \( \frac{1}{\text{time}} \)
rate = \( \frac{1}{50} \)
rate = 0.020 s\(^{-1}\)
Example 2:
Reaction A takes 20 s and reaction B takes 80 s. Compare their rates.
▶️ Answer/Explanation
Rate A = \( \frac{1}{20} = 0.050 \) s\(^{-1}\)
Rate B = \( \frac{1}{80} = 0.0125 \) s\(^{-1}\)
Reaction A has a higher rate.
Therefore, reaction A is faster than reaction B.
9.3(ii) Calculating Rate of Reaction from a Graph
The rate of a reaction can also be determined from the gradient of a suitable graph, such as a graph of concentration, volume of gas, or mass against time.
Rate from Gradient
The rate of reaction is equal to the gradient (slope) of the graph.
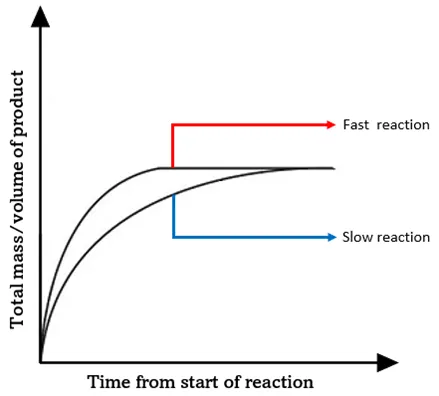
- A steeper gradient indicates a faster rate of reaction.
- A less steep gradient indicates a slower rate of reaction.
The gradient can be determined by drawing a tangent to the curve.
Initial Rate
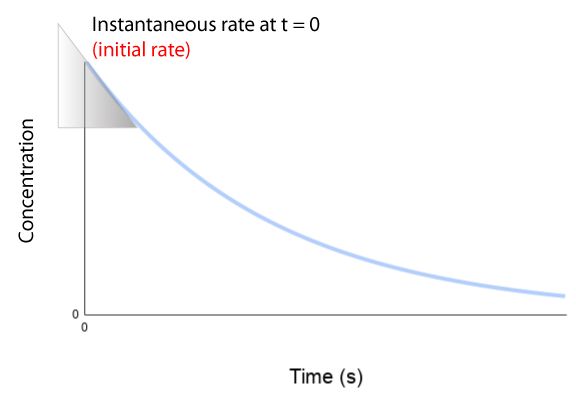
- The initial rate is the rate at the very start of the reaction (at time = 0).
- It is found by drawing a tangent at the beginning of the curve.
- The gradient of this tangent gives the initial rate.
The initial rate is usually the highest because reactant concentration is greatest at the start.
Rate at a Specific Time, t
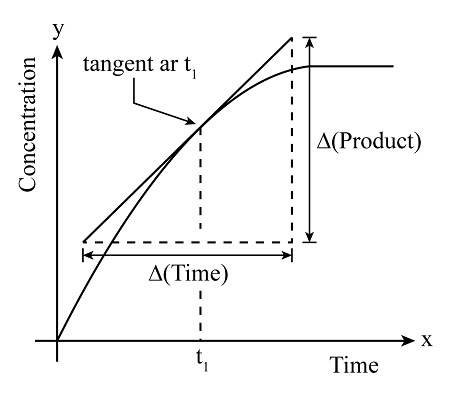
- The rate at a specific time is found by drawing a tangent at that point on the curve.
- The gradient of the tangent gives the rate at that time.
- As time increases, the gradient usually decreases because reactants are being used up.
Calculating the Gradient
- Choose two points on the tangent line.
- Calculate the change in the y-axis value (e.g. concentration or volume).
- Calculate the change in time (x-axis).
- Use: gradient = \( \frac{\Delta y}{\Delta x} \)
Therefore, the gradient of the tangent represents the rate of reaction at that point.
Example 1:
Explain how to determine the initial rate from a graph of volume of gas against time.
▶️ Answer/Explanation
Draw a tangent to the curve at time = 0.
Choose two points on the tangent line.
Calculate the gradient using \( \frac{\Delta y}{\Delta x} \).
This gradient represents the initial rate of reaction.
Example 2:
Explain why the rate of reaction decreases over time based on the graph.
▶️ Answer/Explanation
As the reaction proceeds, reactants are used up.
The concentration of reactants decreases.
This leads to fewer collisions between particles.
Therefore, the gradient becomes less steep and the rate decreases.
