Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.4 Maxwell–Boltzmann distribution-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.4 Maxwell–Boltzmann distribution- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.4 Maxwell–Boltzmann distribution- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.4 understand qualitatively, in terms of the Maxwell–Boltzmann distribution of molecular energies, how changes in temperature affect the rate of a reaction
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.4 Maxwell–Boltzmann Distribution and Temperature
The effect of temperature on the rate of a reaction can be explained using the Maxwell–Boltzmann distribution of molecular energies.
Maxwell–Boltzmann Distribution
The Maxwell–Boltzmann distribution shows how the energies of particles in a system are distributed at a given temperature.
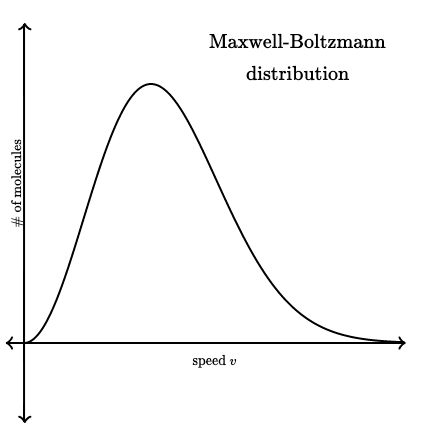
- Not all particles have the same energy.
- Most particles have intermediate energies.
- A small number of particles have very high or very low energies.
The area under the curve represents the total number of particles.
Activation Energy and the Distribution
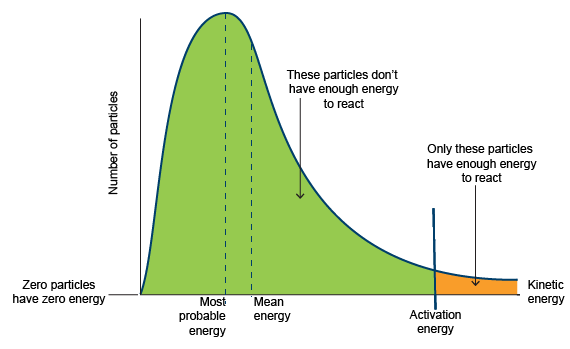
- The activation energy is the minimum energy required for a reaction to occur.
- Only particles with energy greater than or equal to the activation energy can react.
- On the distribution graph, this corresponds to the area to the right of the activation energy line.
This area represents the proportion of particles able to undergo successful collisions.
Effect of Increasing Temperature
- Increasing temperature increases the average kinetic energy of particles.
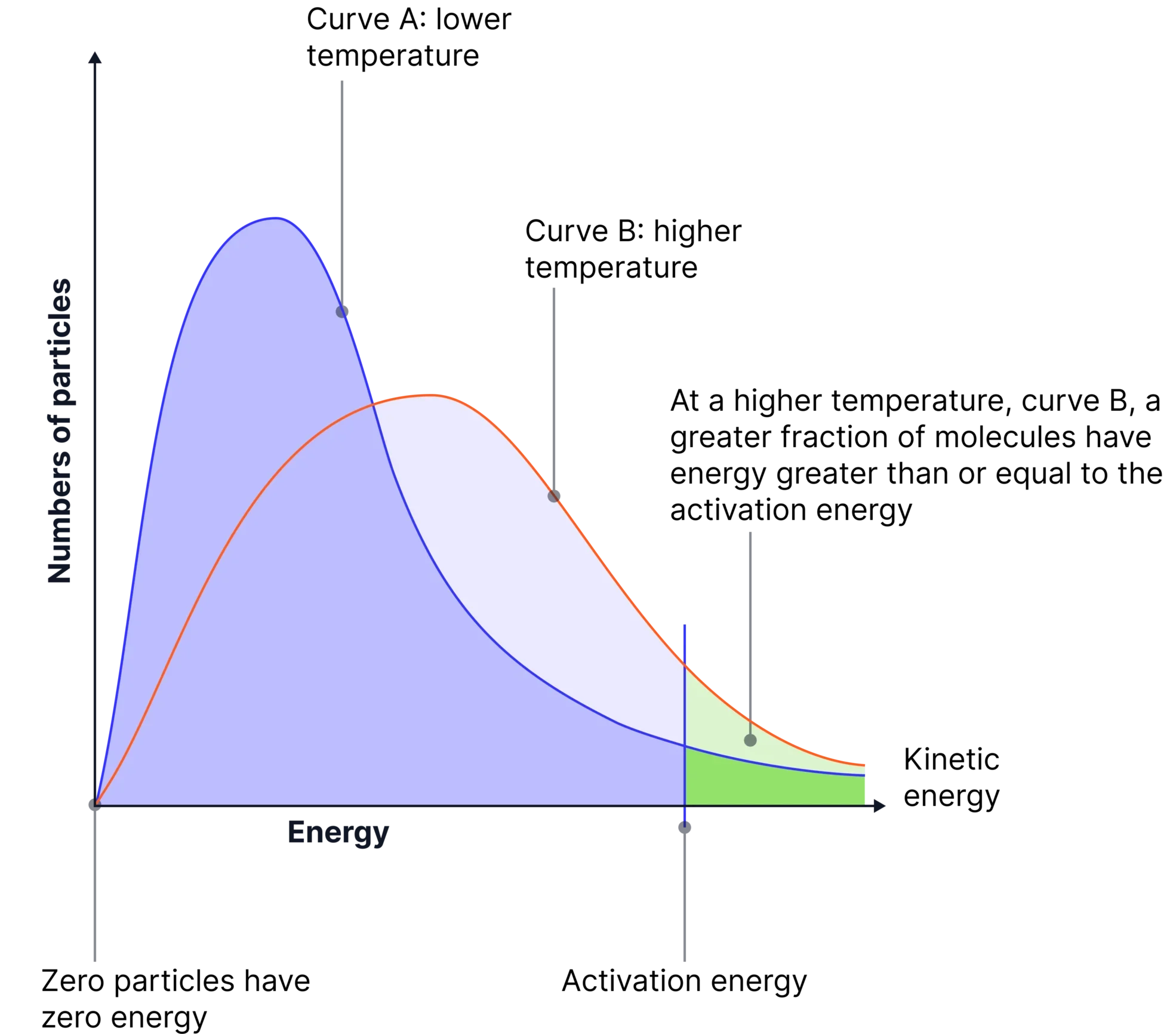
- The distribution curve becomes broader and flatter.
- The peak of the curve decreases and shifts to higher energy.
- A much larger proportion of particles have energy greater than the activation energy.
Therefore, the number of successful collisions increases significantly.
This explains why even a small increase in temperature leads to a large increase in the rate of reaction.
Effect of Decreasing Temperature
- Decreasing temperature lowers the kinetic energy of particles.
- The distribution curve becomes narrower and taller.
- Fewer particles have energy greater than the activation energy.
- The number of successful collisions decreases.
Therefore, the rate of reaction decreases.
Key Explanation
- Temperature does not just increase collision frequency.
- It increases the proportion of particles with sufficient energy.
- This leads to a significant increase in successful collisions.
Therefore, temperature has a large effect on reaction rate because it increases both collision frequency and the number of successful collisions.
Example 1:
Explain, using the Maxwell–Boltzmann distribution, why increasing temperature increases the rate of reaction.
▶️ Answer/Explanation
Increasing temperature causes the distribution curve to become broader and shift to higher energy.
A greater proportion of particles have energy greater than the activation energy.
Therefore, more particles can undergo successful collisions.
This increases the rate of reaction.
Example 2:
On a Maxwell–Boltzmann graph, explain what the area to the right of the activation energy represents.
▶️ Answer/Explanation
The area represents the proportion of particles with energy greater than or equal to the activation energy.
These particles are able to undergo successful collisions.
Therefore, this area represents the fraction of particles that can react.
