Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.5 Catalysts and activation energy-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.5 Catalysts and activation energy- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.5 Catalysts and activation energy- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.5 understand the role of catalysts in providing alternative reaction routes of lower activation energy
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.5 Catalysts and Activation Energy
Catalysts increase the rate of a chemical reaction by providing an alternative reaction pathway.
Definition of a Catalyst
A catalyst is a substance that increases the rate of a reaction without being used up in the process. 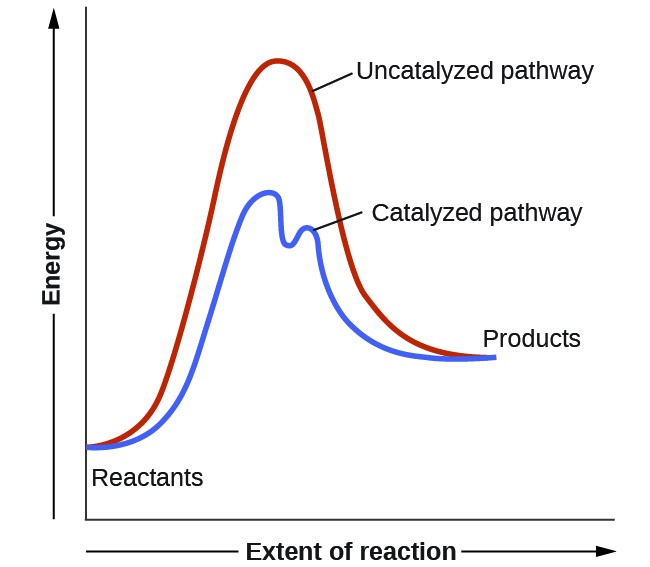
Alternative Reaction Pathway
A catalyst provides an alternative route for the reaction to occur.
- This alternative pathway has a lower activation energy.
- As a result, more particles have sufficient energy to react.
- Therefore, the number of successful collisions increases.
This leads to an increase in the rate of reaction.
Effect on Activation Energy
The catalyst lowers the activation energy required for the reaction.
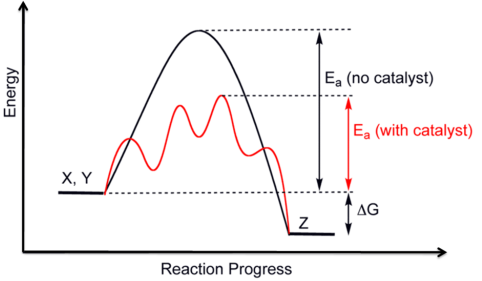
- This means a greater proportion of particles can overcome the energy barrier.
- The overall energy change of the reaction remains unchanged.
Therefore, the catalyst affects the rate of reaction but does not affect the enthalpy change.
Key Points about Catalysts
- Catalysts are not consumed and remain chemically unchanged at the end of the reaction.
- Only a small amount of catalyst is needed.
- Catalysts speed up both the forward and reverse reactions equally.
- Catalysts do not change the position of equilibrium.
Energy Profile Explanation
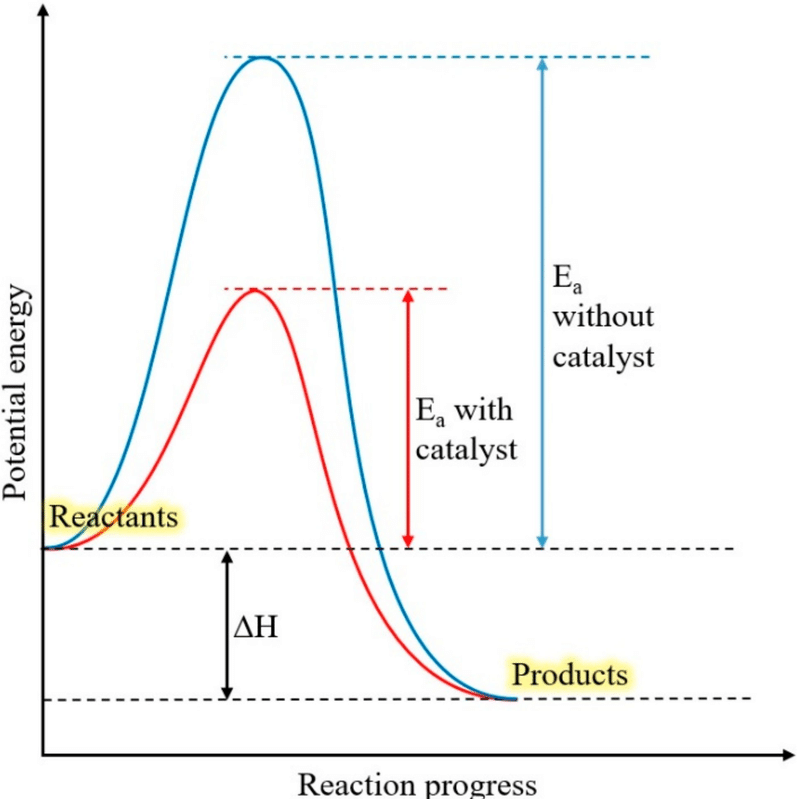
- On an energy profile diagram, the catalysed pathway has a lower peak than the uncatalysed pathway.
- This lower peak represents the reduced activation energy.
- The energy difference between reactants and products remains the same.
Therefore, catalysts increase the rate by lowering activation energy without changing the overall energy change of the reaction.
Example 1:
Explain how a catalyst increases the rate of a reaction.
▶️ Answer/Explanation
A catalyst provides an alternative reaction pathway with lower activation energy.
More particles have sufficient energy to react.
Therefore, the number of successful collisions increases.
This increases the rate of reaction.
Example 2:
Explain why a catalyst does not change the enthalpy change of a reaction.
▶️ Answer/Explanation
A catalyst provides an alternative pathway but does not change the energy of reactants or products.
The energy difference between reactants and products remains the same.
Therefore, the enthalpy change is unchanged.
