Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.7 Catalysts in industry-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.7 Catalysts in industry- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.7 Catalysts in industry- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.7 understand the use of catalysts in industry to make processes more sustainable by using less energy and/or higher atom economy
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.7 Catalysts and Sustainability in Industry
Catalysts are widely used in industry to make chemical processes more sustainable by reducing energy requirements and improving atom economy.
Reducing Energy Requirements
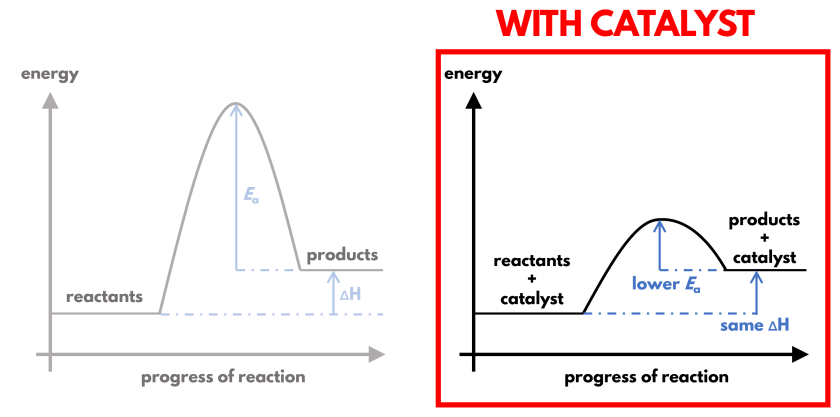
- Catalysts provide an alternative reaction pathway with lower activation energy.
- This allows reactions to occur at lower temperatures and sometimes lower pressures.
- Less energy is required to carry out the reaction.
- This reduces the use of fossil fuels and lowers energy costs.
Therefore, catalysts make processes more energy-efficient and environmentally friendly.
Improving Atom Economy
- Catalysts can increase the selectivity of a reaction.
- This means more of the desired product is formed and fewer unwanted by-products are produced.
- As a result, a greater proportion of reactant atoms are incorporated into the desired product.
- This improves the atom economy of the process.
Higher atom economy means less waste is produced, making the process more sustainable.
Economic and Environmental Benefits
- Lower energy use reduces production costs.
- Reduced waste lowers disposal costs and environmental impact.
- More efficient use of raw materials conserves resources.
Therefore, catalysts play a key role in developing sustainable industrial processes.
Key Idea
- Catalysts do not just increase rate.
- They make reactions more efficient, economical, and environmentally friendly.
Therefore, catalysts are essential in green chemistry and sustainable industrial design.
Example 1:
Explain how catalysts make industrial processes more sustainable.
▶️ Answer/Explanation
Catalysts lower the activation energy of reactions.
This allows reactions to occur at lower temperatures, reducing energy use.
Catalysts also increase selectivity, producing fewer by-products.
Therefore, less waste is formed and the process becomes more sustainable.
Example 2:
Explain why higher atom economy is important in industrial processes.
▶️ Answer/Explanation
Higher atom economy means more reactant atoms are converted into useful products.
Less waste is produced.
This reduces environmental impact and disposal costs.
Therefore, it makes the process more sustainable and efficient.
