Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 9.9 Dynamic equilibrium-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.9 Dynamic equilibrium- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 9.9 Dynamic equilibrium- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
9.9 know that many reactions are readily reversible and that they can reach a state of dynamic equilibrium in which:
i the rate of the forward reaction is equal to the rate of the backward reaction
ii the concentrations of the reactants and the products remain constant
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
9.9 Dynamic Equilibrium
(i): Equality of Forward and Backward Reaction Rates
Many chemical reactions are reversible, meaning they can proceed in both the forward and backward directions. In a closed system, such reactions can reach a state known as dynamic equilibrium, where both reactions continue to occur but with no overall change in the system.
Dynamic Equilibrium (Rate Perspective)
At dynamic equilibrium, the rate of the forward reaction is equal to the rate of the backward reaction.
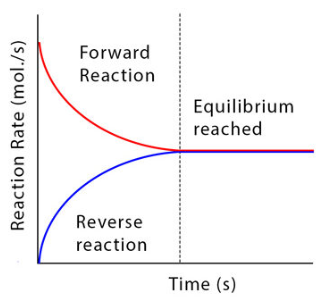
Initially, when reactants are present and products are absent, only the forward reaction occurs at a significant rate. As products begin to form, the backward reaction starts to occur. Over time:
- The rate of the forward reaction decreases as reactant concentration decreases.
- The rate of the backward reaction increases as product concentration increases.
- Eventually, both rates become equal, establishing dynamic equilibrium.
At this point, reactions are still occurring in both directions, but there is no net change in the amounts of reactants and products because they are being converted at equal rates. This is why the equilibrium is described as dynamic rather than static.
Illustration with a Reversible Reaction
\( \mathrm{N_2(g) + 3H_2(g) \ \rightleftharpoons \ 2NH_3(g)} \)
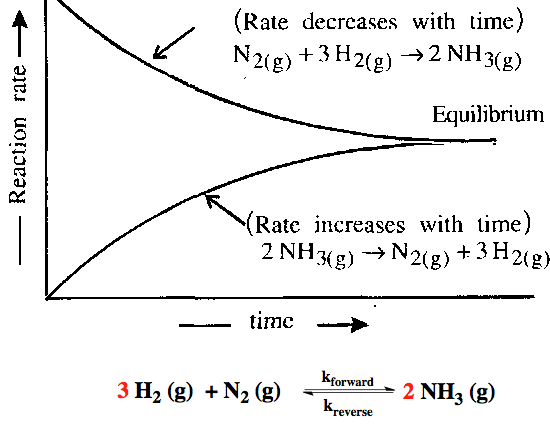
In this system:
- The forward reaction forms \( \mathrm{NH_3} \) from \( \mathrm{N_2} \) and \( \mathrm{H_2} \).
- The backward reaction decomposes \( \mathrm{NH_3} \) back into \( \mathrm{N_2} \) and \( \mathrm{H_2} \).
- At equilibrium, both processes occur at equal rates, so there is no overall change in composition.
Key Features of Equal Reaction Rates
- The equality of rates means there is no net change in the system at equilibrium.
- Both forward and backward reactions continue simultaneously.
- The system must be closed to prevent loss of reactants or products.
- The dynamic nature arises because particles are continuously reacting, even though macroscopic properties remain unchanged.
Example 1:
Explain what is meant by the statement “the rate of the forward reaction is equal to the rate of the backward reaction” at dynamic equilibrium.
▶️ Answer/Explanation
At dynamic equilibrium, the forward and backward reactions occur at the same rate, meaning reactants are converted into products at the same rate as products are converted back into reactants.
As a result, there is no net change in the amounts of substances present, even though both reactions continue to occur.
This demonstrates that equilibrium is dynamic, not static.
Example 2:
In a reversible reaction, explain why the rates of the forward and backward reactions eventually become equal.
▶️ Answer/Explanation
Initially, only the forward reaction occurs at a significant rate because only reactants are present.
As products form, the backward reaction begins and its rate increases due to increasing product concentration.
At the same time, the rate of the forward reaction decreases as reactant concentration decreases.
Eventually, the two rates become equal, establishing dynamic equilibrium where both reactions continue but with no net change.
(ii): Constant Concentrations of Reactants and Products
In a reversible reaction at dynamic equilibrium, the concentrations of reactants and products remain constant over time. This does not mean the reactions have stopped, but rather that the system has reached a balance due to equal rates of opposing reactions.
Constant Concentrations at Equilibrium
At dynamic equilibrium, the concentrations of reactants and products remain constant because they are being formed and consumed at equal rates.
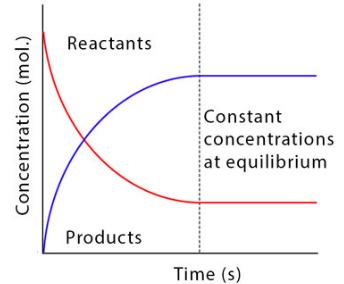
As the reaction proceeds towards equilibrium:
- The concentration of reactants decreases as they are converted into products.
- The concentration of products increases as they are formed.
- Eventually, a point is reached where both concentrations stop changing.
This occurs because:
- The rate of the forward reaction equals the rate of the backward reaction.
- The amount of reactant used per unit time equals the amount regenerated.
- The amount of product formed per unit time equals the amount decomposed.
Therefore, although particles continue to react, there is no overall change in concentration, making the equilibrium appear constant on a macroscopic scale.
Illustration with a Reversible Reaction
\( \mathrm{N_2(g) + 3H_2(g) \ \rightleftharpoons \ 2NH_3(g)} \)
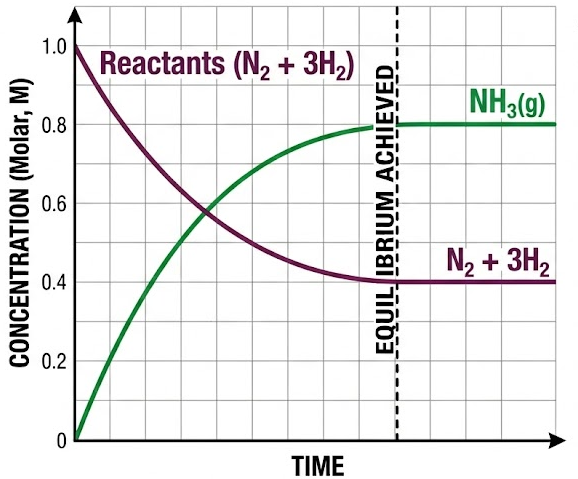
In this system:
- The concentrations of \( \mathrm{N_2} \), \( \mathrm{H_2} \), and \( \mathrm{NH_3} \) become constant at equilibrium.
- However, the forward and backward reactions continue to occur simultaneously.
- The constant concentrations are a direct result of equal reaction rates.
Key Features of Constant Concentrations
- Concentrations remain constant, but not necessarily equal to each other.
- The system must be closed to maintain constant concentrations.
- The equilibrium is dynamic because reactions continue at the particle level.
- Constant concentration is a consequence of equal forward and backward reaction rates.
Example 1:
Explain why the concentrations of reactants and products remain constant at dynamic equilibrium.
▶️ Answer/Explanation
At dynamic equilibrium, the rate of the forward reaction is equal to the rate of the backward reaction.
This means reactants are converted into products at the same rate as products are converted back into reactants.
As a result, there is no net change in the amounts of substances present, so their concentrations remain constant.
Example 2:
A reaction mixture at equilibrium shows constant concentrations of all species. Explain why this does not mean the reaction has stopped.
▶️ Answer/Explanation
Although the concentrations remain constant, both the forward and backward reactions are still occurring.
The constant concentrations indicate that the rates of the two reactions are equal, not that they have stopped.
Therefore, particles continue to react at the microscopic level, making the equilibrium dynamic rather than static.
