Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 4 - 11.1 Kinetics Definitions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 4 – 11.1 Kinetics Definitions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 4 – 11.1 Kinetics Definitions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
11.1 understand the terms:
i rate of reaction
ii rate equation, rate=k[A]m[B]n where m and n are 0, 1 or 2
iii order with respect to a substance in a rate equation
iv overall order of a reaction
v rate constant
vi half-life
vii rate-determining step
viii activation energy
ix heterogeneous and homogeneous catalyst
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
11.1 Chemical Kinetics – Definitions
(i) Rate of Reaction
The rate of reaction is the change in concentration of a reactant or product per unit time.

- Can be measured as decrease in reactant concentration or increase in product concentration
- Units: \( \mathrm{mol\ dm^{-3}\ s^{-1}} \)
- Rate is highest at the start of the reaction
- Rate decreases as reactants are used up
(ii) Rate Equation
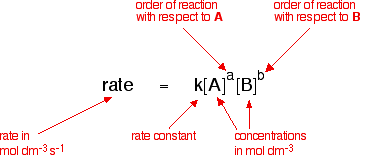
The rate equation shows how the rate of a reaction depends on the concentrations of reactants.
\( \mathrm{rate = k[A]^m[B]^n} \)
- \( \mathrm{k} \) = rate constant
- \( \mathrm{[A], [B]} \) = concentrations of reactants
- \( \mathrm{m, n} \) = orders (0, 1 or 2)
- Determined experimentally (not from balanced equation)
(iii) Order with Respect to a Substance
The order with respect to a substance is the power to which its concentration is raised in the rate equation.
- Indicates how strongly a reactant affects the rate
- Determined experimentally
Types:
- Zero order: rate independent of concentration
- First order: rate ∝ concentration
- Second order: rate ∝ (concentration)²

Example 1:
The rate equation for a reaction is \( \mathrm{rate = k[A]^2[B]^0} \). Explain the order with respect to each reactant and describe how the rate changes when the concentration of each reactant is doubled.
▶️ Answer/Explanation
Order with respect to \( \mathrm{A} \) = 2
Order with respect to \( \mathrm{B} \) = 0
Doubling \( \mathrm{[A]} \):
Rate increases by \( 2^2 = 4 \) times
Doubling \( \mathrm{[B]} \):
No change in rate (zero order)
This shows that only \( \mathrm{A} \) affects the rate of reaction.
Example 2:
In an experiment, increasing the concentration of reactant A doubles the rate, while increasing the concentration of B has no effect. Deduce the rate equation and explain the order with respect to each reactant.
▶️ Answer/Explanation
Doubling \( \mathrm{[A]} \) doubles rate → first order in A
Changing \( \mathrm{[B]} \) has no effect → zero order in B
Rate equation:
\( \mathrm{rate = k[A]^1[B]^0} \)
This means:
Rate depends only on concentration of A
B does not influence the rate
(iv) Overall Order of a Reaction
The overall order of a reaction is the sum of the powers of the concentrations of all reactants in the rate equation.

- Obtained by adding individual orders
- Example: \( \mathrm{rate = k[A]^2[B]} \) → overall order = 3
- Shows overall dependence of rate on concentration
- Used to determine units of \( \mathrm{k} \)
- Determined experimentally
(v) Rate Constant (\( \mathrm{k} \))
The rate constant is the proportionality constant in the rate equation that relates the rate of reaction to the concentrations of reactants.

- Constant for a given reaction at fixed temperature
- Value changes with temperature and catalysts
- Larger \( \mathrm{k} \) → faster reaction
- Units depend on overall order of reaction
(vi) Half-life
The half-life is the time taken for the concentration of a reactant to decrease to half of its initial value.

- For first-order reactions: half-life is constant
- Independent of initial concentration (first order only)
- For zero and second order: half-life changes during reaction
- Used to identify reaction order experimentally
Example 1:
A reaction follows the rate equation \( \mathrm{rate = k[A]^2[B]} \). Determine the overall order and deduce the units of \( \mathrm{k} \).
▶️ Answer/Explanation
Order with respect to A = 2
Order with respect to B = 1
Overall order = 3
Units of rate = \( \mathrm{mol\ dm^{-3}\ s^{-1}} \)
\( \mathrm{k = \frac{rate}{[A]^2[B]}} \)
Units of \( \mathrm{k} \):
\( \mathrm{mol\ dm^{-3}\ s^{-1} \div (mol\ dm^{-3})^3} \)
= \( \mathrm{dm^6\ mol^{-2}\ s^{-1}} \)
Example 2:
A reaction shows half-lives of 40 s, 40 s and 40 s as concentration decreases. Explain what this indicates about the reaction and the rate constant.
▶️ Answer/Explanation
Constant half-life → reaction is first order
For first-order reactions:
Rate ∝ concentration
Half-life is independent of concentration
The rate constant \( \mathrm{k} \) remains constant (at constant temperature)
(vii) Rate-Determining Step
The rate-determining step is the slowest step in a multi-step reaction mechanism and controls the overall rate of the reaction.
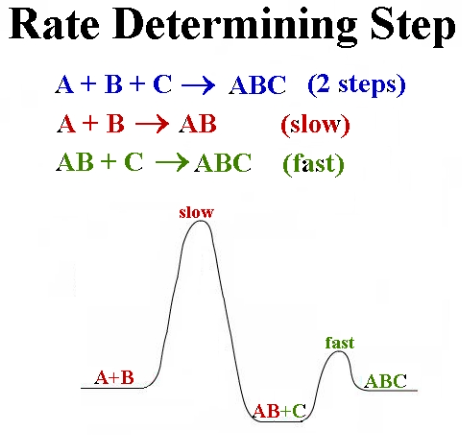
- Acts as a bottleneck in the reaction pathway
- Determines the overall rate of the reaction
- Rate equation is usually based on reactants in this step
- Faster steps do not affect the overall rate significantly
Energy Profile Explanation:
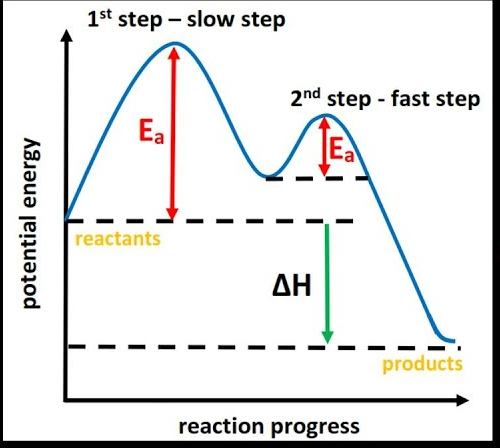
- Reactions may occur in multiple steps, each with its own activation energy
- The step with the highest activation energy (\( \mathrm{E_a} \)) is the slowest
- Therefore, this step is the rate-determining step
- The overall rate depends on how fast this step occurs
- Energy profile diagrams show this as the highest peak
(viii) Activation Energy (\( \mathrm{E_a} \))
Activation energy is the minimum energy that reacting particles must possess for a successful collision to occur.
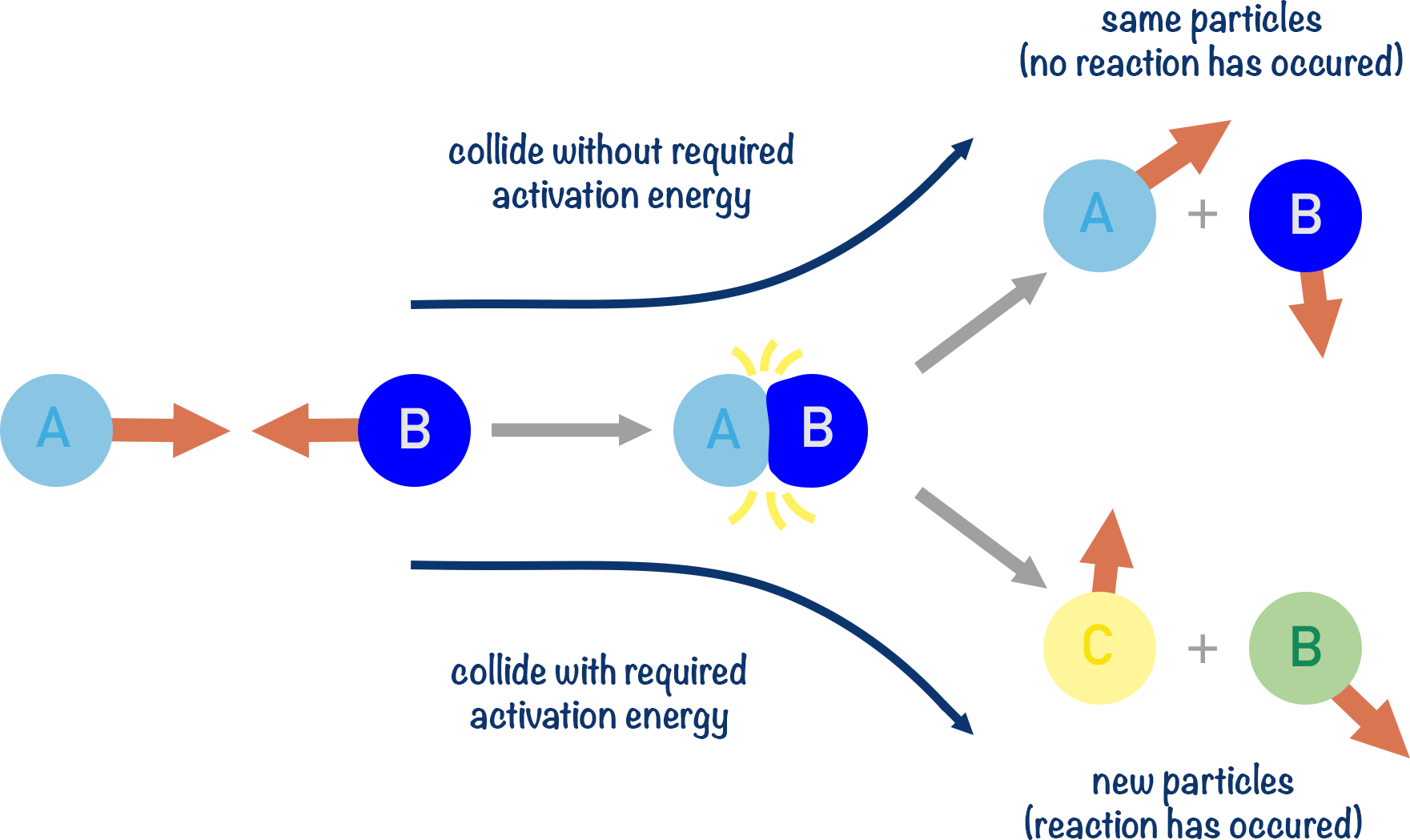
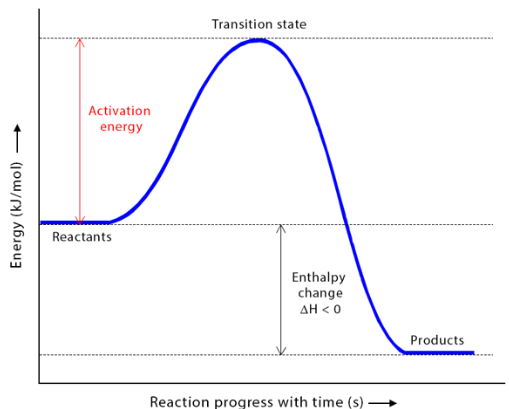
- Represents the energy barrier for a reaction
- Only particles with energy ≥ \( \mathrm{E_a} \) can react
- Higher \( \mathrm{E_a} \) → slower reaction
- Increasing temperature increases number of particles with sufficient energy
- Catalysts lower activation energy
(ix) Homogeneous and Heterogeneous Catalysts
Catalysts are substances that increase the rate of a reaction without being consumed and can be classified based on their phase relative to the reactants.
- Homogeneous catalyst: same phase as reactants
- Reaction occurs throughout the mixture
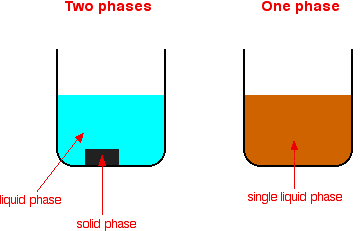
- Heterogeneous catalyst: different phase from reactants
- Reaction occurs on the surface of the catalyst
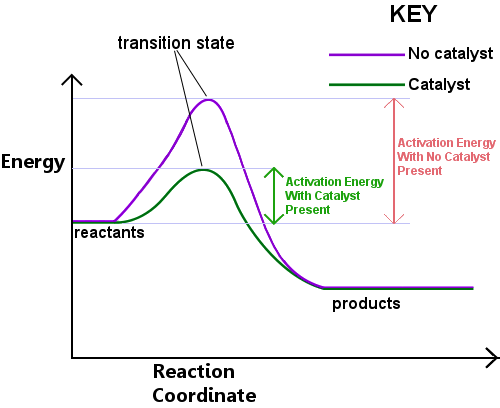
- Provide alternative pathway with lower activation energy
- Not consumed in the reaction
- Increase rate of both forward and reverse reactions
Example 1:
A reaction mechanism consists of two steps: Step 1 (slow): \( \mathrm{2A + B \rightarrow C} \) Step 2 (fast): \( \mathrm{C \rightarrow D} \) Deduce the rate equation and identify the rate-determining step.
▶️ Answer/Explanation
Slow step = Step 1 → rate-determining step
Rate depends on reactants in slow step:
\( \mathrm{rate = k[A]^2[B]} \)
Step 2 is fast and does not affect rate
Example 2:
Explain why increasing temperature increases reaction rate in terms of activation energy and particle energy distribution.
▶️ Answer/Explanation
Increasing temperature increases kinetic energy of particles
More particles have energy ≥ \( \mathrm{E_a} \)
This increases number of successful collisions
Therefore, reaction rate increases
