Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 4 - 11.11 Heterogeneous catalysis-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 4 – 11.11 Heterogeneous catalysis- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 4 – 11.11 Heterogeneous catalysis- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
11.11 understand the use of a solid (heterogeneous) catalyst for industrial reactions, in the gas phase, in terms of providing a surface for the reaction
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
11.11 Heterogeneous Catalysts: Surface for Gas-Phase Reactions
In many industrial processes, reactions occur in the gas phase using a solid catalyst. Such catalysts are described as heterogeneous because they are in a different physical state from the reactants. Their effectiveness arises from their ability to provide a surface on which reactant molecules can adsorb, react, and then desorb as products.
Heterogeneous Catalyst
A heterogeneous catalyst is a catalyst in a different physical state from the reactants, typically a solid catalyst with gaseous reactants.
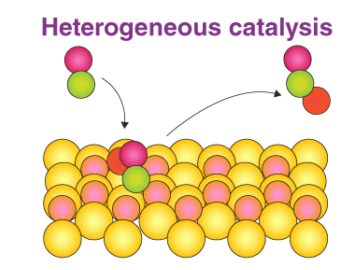
Role of the Catalyst Surface
The solid catalyst provides an active surface where reactions occur. The mechanism involves several key steps:
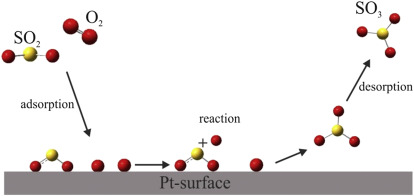
- Adsorption: Reactant molecules attach to the catalyst surface at active sites.
- Bonds within reactant molecules may be weakened due to interactions with the surface.
- Reaction on the surface: Adsorbed species react with each other more readily because they are held close together and properly oriented.
- Desorption: Product molecules leave the surface, freeing active sites for further reactions.
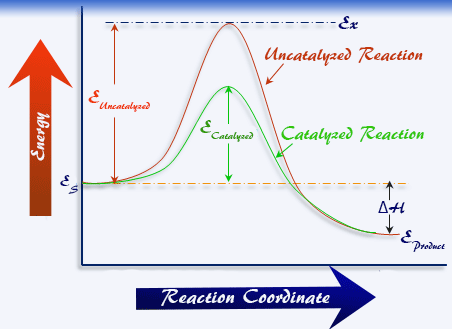
This process lowers the activation energy by providing an alternative reaction pathway, increasing the rate of reaction without the catalyst being consumed.
Importance of Surface Area
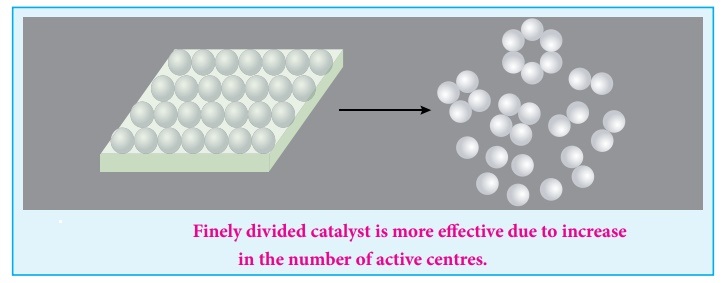
- Catalysts are often finely divided to increase surface area.
- A larger surface area provides more active sites for adsorption.
- This increases the frequency of successful collisions and the overall reaction rate.
Illustration: Haber Process
\( \mathrm{N_2(g) + 3H_2(g) \ \rightleftharpoons \ 2NH_3(g)} \)
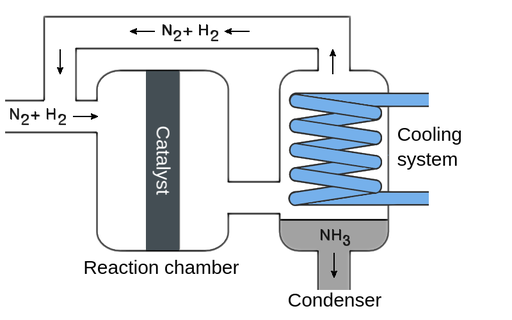
- Iron is used as a solid catalyst.
- \( \mathrm{N_2} \) and \( \mathrm{H_2} \) molecules adsorb onto the iron surface.
- The strong triple bond in \( \mathrm{N_2} \) is weakened on the surface.
- Hydrogen atoms combine with nitrogen atoms to form \( \mathrm{NH_3} \).
- \( \mathrm{NH_3} \) molecules desorb from the surface.
Key Features
- Provides an alternative reaction pathway with lower activation energy.
- Involves adsorption, surface reaction, and desorption.
- Requires a solid surface with active sites.
- Not consumed during the reaction.
Example 1:
Explain how a solid catalyst increases the rate of a gas-phase reaction.
▶️ Answer/Explanation
A solid catalyst provides a surface on which reactant molecules can adsorb at active sites.
Adsorption weakens bonds within the reactant molecules and brings them into close proximity.
This increases the likelihood of successful collisions and provides an alternative reaction pathway with lower activation energy.
As a result, the rate of reaction increases.
Example 2:
Explain why finely divided catalysts are more effective in industrial reactions.
▶️ Answer/Explanation
Finely divided catalysts have a larger surface area compared to larger pieces of the same material.
This provides more active sites for adsorption of reactant molecules.
As more molecules can adsorb simultaneously, the frequency of successful reactions increases.
Therefore, the overall rate of reaction is higher.
