Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 4 - 11.7 Rate-determining step from rate equation-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 4 – 11.7 Rate-determining step from rate equation- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 4 – 11.7 Rate-determining step from rate equation- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
11.7 be able to deduce the rate-determining step from a rate equation and vice versa
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
11.7 Deducing the Rate-Determining Step and Rate Equation
The rate equation and the rate-determining step are closely related, as the slowest step in a mechanism controls the overall rate of the reaction.
From Rate Equation → Rate-Determining Step
The species appearing in the rate equation are those involved in the rate-determining (slow) step.
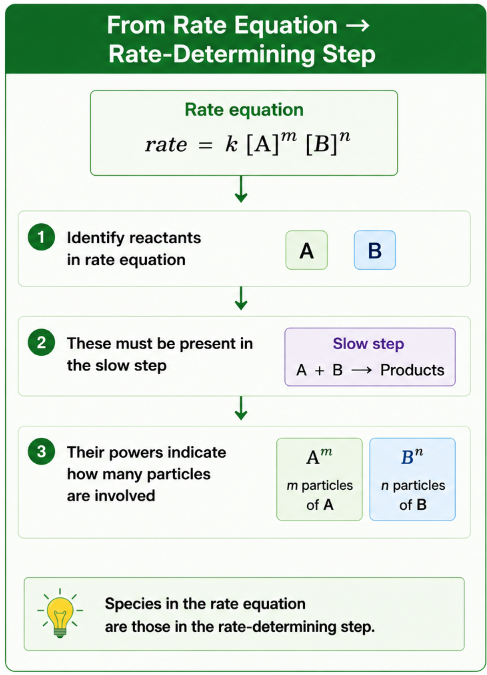
- Identify reactants in rate equation
- These must be present in the slow step
- Their powers indicate how many particles are involved
From Mechanism → Rate Equation
The rate equation is determined by the reactants in the slow (rate-determining) step of the mechanism.
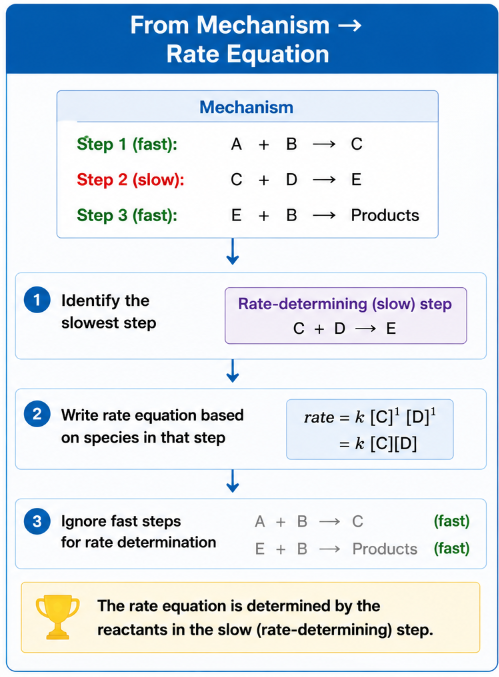
- Identify the slowest step
- Write rate equation based on species in that step
- Ignore fast steps for rate determination
Key Rules
- Rate equation must match the slow step
- Species not in slow step do not appear in rate equation
- Intermediates do not appear in overall rate equation
- Powers in rate equation reflect molecularity of slow step
Key Insight
- Rate-determining step controls the overall reaction rate
- Fast steps occur after or before but do not affect rate
Example 1
The rate equation for a reaction is:
\( \mathrm{rate = k[A]^2[B]} \)
Suggest a possible rate-determining step.
▶️ Answer/Explanation
Slow step must involve A and B
Powers indicate:
2 molecules of A and 1 molecule of B
Step: \( \mathrm{2A + B \rightarrow intermediate} \)
This matches rate equation
Example 2
A reaction mechanism is:
Step 1 (slow): \( \mathrm{A + B \rightarrow X} \) Step 2 (fast): \( \mathrm{X + C \rightarrow products} \)
Deduce the rate equation.
▶️ Answer/Explanation
Slow step determines rate
Reactants in slow step: A and B
\( \mathrm{rate = k[A][B]} \)
C does not appear as it is in fast step
