Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 4 - 12.13 Born-Haber cycles-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 4 – 12.13 Born-Haber cycles- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 4 – 12.13 Born-Haber cycles- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
Update
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
12.13 Born–Haber Cycles and Related Calculations
A Born–Haber cycle is an energy cycle used to calculate lattice energy or other unknown enthalpy changes in ionic compounds. It applies Hess’s Law by breaking the formation of an ionic solid into a series of steps involving atoms, ions and electrons.
Key Idea (Hess’s Law)
The total enthalpy change is independent of the pathway taken, so the sum of enthalpy changes in a cycle is zero.
Typical Steps in a Born–Haber Cycle
For an ionic compound such as \( \mathrm{NaCl} \):
\( \mathrm{Na(s) + \frac{1}{2}Cl_2(g) \rightarrow NaCl(s)} \)
The cycle includes:
- Enthalpy of formation (\( \mathrm{\Delta_f H} \))
- Atomisation of metal: \( \mathrm{Na(s) \rightarrow Na(g)} \)
- Atomisation of non-metal: \( \mathrm{\frac{1}{2}Cl_2(g) \rightarrow Cl(g)} \)
- Ionisation energy: \( \mathrm{Na(g) \rightarrow Na^+(g) + e^-} \)
- Electron affinity: \( \mathrm{Cl(g) + e^- \rightarrow Cl^-(g)} \)
- Lattice energy: \( \mathrm{Na^+(g) + Cl^-(g) \rightarrow NaCl(s)} \)
Energy Cycle Relationship
\( \mathrm{\Delta_f H = \Delta_{at}H (metal) + \Delta_{at}H (non-metal) + IE + EA + \Delta_{latt}H} \)
Rearranged to find lattice energy:
\( \mathrm{\Delta_{latt}H = \Delta_f H – (other\ enthalpy\ changes)} \)
Important Notes
- Pay attention to signs (endothermic = positive, exothermic = negative).
- Include all steps: atomisation, ionisation, electron affinity.
- Multiply values according to stoichiometry.
Diagrammatic Representation (Concept)
The Born–Haber cycle is typically drawn as an energy level diagram showing:
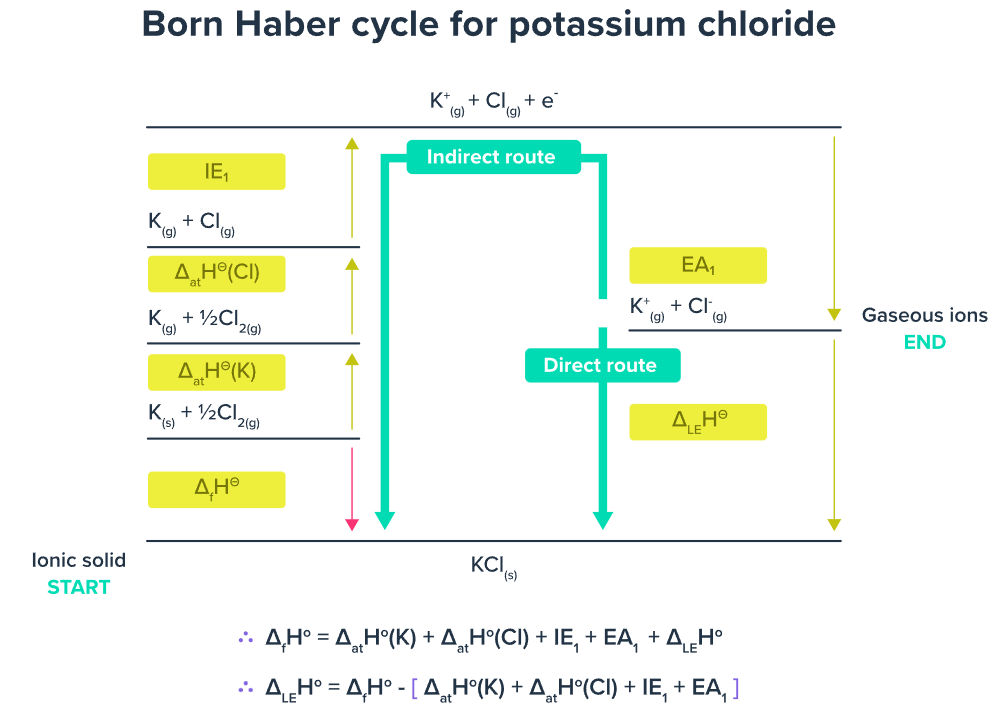
- Direct route: elements → ionic solid (enthalpy of formation).
- Indirect route: elements → gaseous atoms → gaseous ions → ionic solid.
Key Features
- Used to calculate lattice energy or unknown enthalpy changes.
- Based on Hess’s Law.
- Involves multiple energy steps (atomisation, IE, EA).
- Requires careful handling of signs and coefficients.
Example 1:
Calculate the lattice energy of \( \mathrm{NaCl} \) given:
- \( \mathrm{\Delta_f H = -411\ kJ\ mol^{-1}} \)
- \( \mathrm{\Delta_{at}H(Na) = +108} \)
- \( \mathrm{\frac{1}{2}Cl_2 \rightarrow Cl = +121} \)
- \( \mathrm{IE(Na) = +496} \)
- \( \mathrm{EA(Cl) = -349} \)
▶️ Answer/Explanation
Sum of steps (excluding lattice energy):
\( \mathrm{108 + 121 + 496 – 349 = 376} \)
Using Hess’s Law:
\( \mathrm{-411 = 376 + \Delta_{latt}H} \)
\( \mathrm{\Delta_{latt}H = -411 – 376 = -787\ kJ\ mol^{-1}} \)
Therefore, lattice energy = \( \mathrm{-787\ kJ\ mol^{-1}} \).
Example 2:
Explain why lattice energy becomes more exothermic as ionic charge increases.
▶️ Answer/Explanation
Lattice energy depends on the electrostatic attraction between oppositely charged ions.
Higher ionic charges result in stronger electrostatic forces.
Stronger attractions release more energy when the lattice forms.
Therefore, lattice energy becomes more exothermic with increasing ionic charge.
