Question 1
B. Hydrolysis
C. Condensation reaction
D. Peptolysis
▶️ Answer/Explanation
Correct Option: B. Hydrolysis
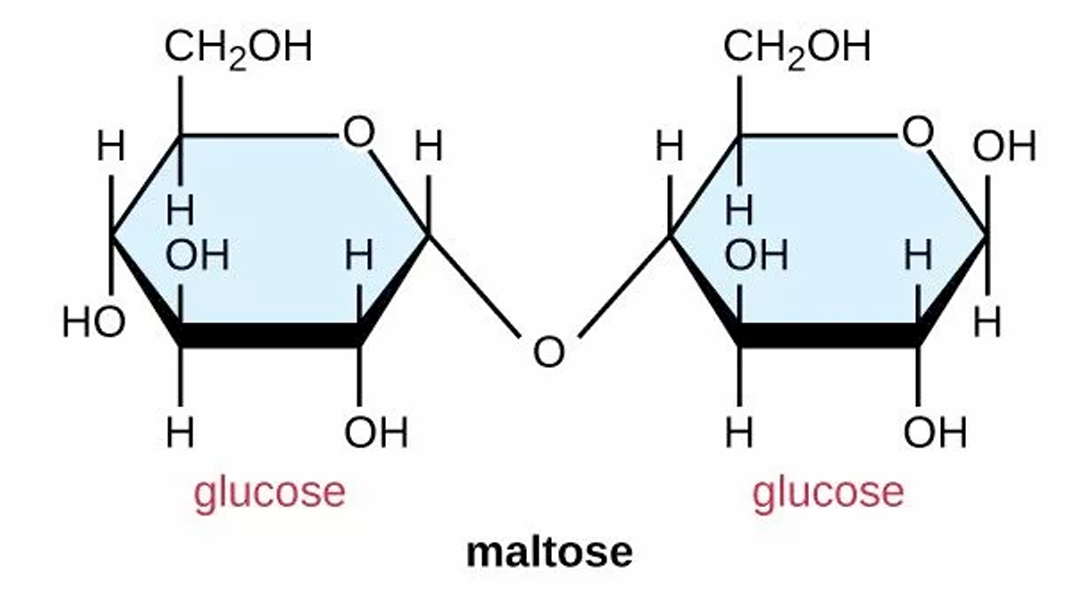
Maltose is a disaccharide with the chemical formula $C_{12}H_{22}O_{11}$.
To break the covalent glycosidic bond, a molecule of $H_{2}O$ must be added.
The term “hydrolysis” literally translates to “water-splitting” ($hydro$ = water, $lysis$ = break).
One glucose molecule receives a hydroxyl group ($-OH$) and the other receives a hydrogen atom ($-H$).
This reaction results in two separate monosaccharides: $C_{12}H_{22}O_{11} + H_{2}O \rightarrow 2C_{6}H_{12}O_{6}$.
Options A and C (dehydration/condensation) are used to build molecules, not break them.
Option D is a distractor term not used in standard carbohydrate chemistry.
Question 2
B. DNA is typically single stranded, while RNA is typically double stranded.
C. RNA is antiparallel, while DNA is a parallel helix.
D. DNA contains deoxyribose sugar, while RNA contains ribose sugar.
▶️ Answer/Explanation
The correct answer is D.
DNA uses the five-carbon sugar deoxyribose, which lacks an oxygen atom on the $2’$ carbon.
RNA uses the five-carbon sugar ribose, which possesses a hydroxyl ($-OH$) group on the $2’$ carbon.
Both DNA and RNA contain phosphate groups, making statement A incorrect.
DNA is typically a double-stranded helix, whereas RNA is usually single-stranded, making B incorrect.
DNA strands are antiparallel ($5’$ to $3’$ and $3’$ to $5’$), making statement C incorrect.
The presence of different sugars is a fundamental structural distinction between these two nucleic acids.
Question 3
B. Adenine $= 22\%$, Cytosine $= 28\%$, and Guanine $= 28\%$
C. Cytosine $= 22\%$, Adenine $= 32\%$, and Guanine $= 32\%$
D. Guanine $= 22\%$, Cytosine $= 28\%$, and Adenine $= 28\%$
▶️ Answer/Explanation
According to Chargaff’s rule, Adenine ($A$) always pairs with Thymine ($T$), so $\%A = \%T$.
Given $T = 22\%$, it follows that $A = 22\%$.
The combined percentage of $A + T = 22\% + 22\% = 44\%$.
The remaining percentage for Cytosine ($C$) and Guanine ($G$) is $100\% – 44\% = 56\%$.
Since $G$ always pairs with $C$, their percentages are equal: $\%G = \%C$.
Therefore, $G = 56\% / 2 = 28\%$ and $C = 56\% / 2 = 28\%$.
The correct option is B.
Question 4
B. The presence of $5’$ to $3’$ antiparallel directionality.
C. The presence of a $5$ carbon pentose sugar in the backbone of the polymer.
D. The presence of uracil nitrogenous bases.
▶️ Answer/Explanation
Correct Option: D
RNA contains the nitrogenous base uracil, whereas DNA contains thymine.
Both DNA and RNA utilize $5$ carbon pentose sugars, but RNA uses ribose and DNA uses deoxyribose.
Double-helix structures and antiparallel directionality are primary characteristics of DNA.
While some RNA can fold into complex shapes, the unique presence of uracil is a definitive chemical marker for RNA.
Options A and B describe traits typically associated with double-stranded DNA.
Option C is incorrect because both types of nucleic acids contain a $5$ carbon pentose sugar.
Question 5
B. The presence of two or more polypeptides connected by $R$-side chain bonding.
C. Structures such as alpha-helices that are made by backbone hydrogen bonding.
D. The order or sequence of amino acids in a polypeptide chain.
▶️ Answer/Explanation
The correct answer is C.
Secondary structure refers to local folding patterns within a polypeptide.
It is stabilized specifically by hydrogen bonds between atoms of the peptide backbone.
Common examples include the $\alpha$-helix and the $\beta$-pleated sheet.
Option A describes tertiary structure involving $R$-group interactions.
Option B describes quaternary structure involving multiple subunits.
Option D describes the primary structure or amino acid sequence.
Question 6
B. Tertiary structure.
C. Quaternary structure.
D. Either tertiary or quaternary structure depending upon if the cysteine—cysteine $R$-side chain bonds are between amino acids in the same polypeptide or two different polypeptides.
▶️ Answer/Explanation
The correct option is D.
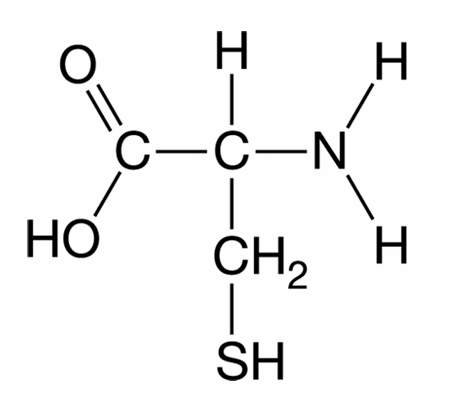
Cysteine contains a sulfhydryl ($-SH$) group in its $R$-group side chain.
Two cysteine side chains can oxidize to form a covalent disulfide bridge ($-S-S-$).
If this bond forms within a single polypeptide chain, it stabilizes the tertiary structure.
If the bond forms between different polypeptide subunits, it stabilizes the quaternary structure.
Secondary structure is primarily stabilized by hydrogen bonds between the backbone, not $R$-groups.
Therefore, the classification depends entirely on whether the bond is intra-chain or inter-chain.
Question 7
▶️ Answer/Explanation
The correct answer is B. Only primary and tertiary structure.
Every protein has a primary structure, which is the unique sequence of amino acids in the polypeptide chain.
Since the protein is described as “highly complex and folded,” it must possess a tertiary structure, which is the overall three-dimensional shape.
The prompt explicitly states there are no alpha ($\alpha$) helices or beta ($\beta$) sheets, which means there is no secondary structure present.
Because it consists of a single polypeptide, it cannot have a quaternary structure, as that requires multiple subunits.
Therefore, the only levels of organization present are the primary and tertiary structures.
Question 8
B. Only secondary structure
C. Only quaternary structure
D. It will not have any level of protein structure
▶️ Answer/Explanation
Denaturation involves the disruption of weak bonds such as hydrogen bonds and ionic interactions.
This process causes the protein to lose its quaternary, tertiary, and secondary structures.
The primary structure consists of amino acids linked by strong covalent peptide bonds.
These covalent bonds are not broken by standard denaturing agents like heat or pH changes.
Therefore, the linear sequence of amino acids remains intact after denaturation.
The protein returns to an unfolded polypeptide chain, retaining only its primary level of organization.
Question 9 – 10
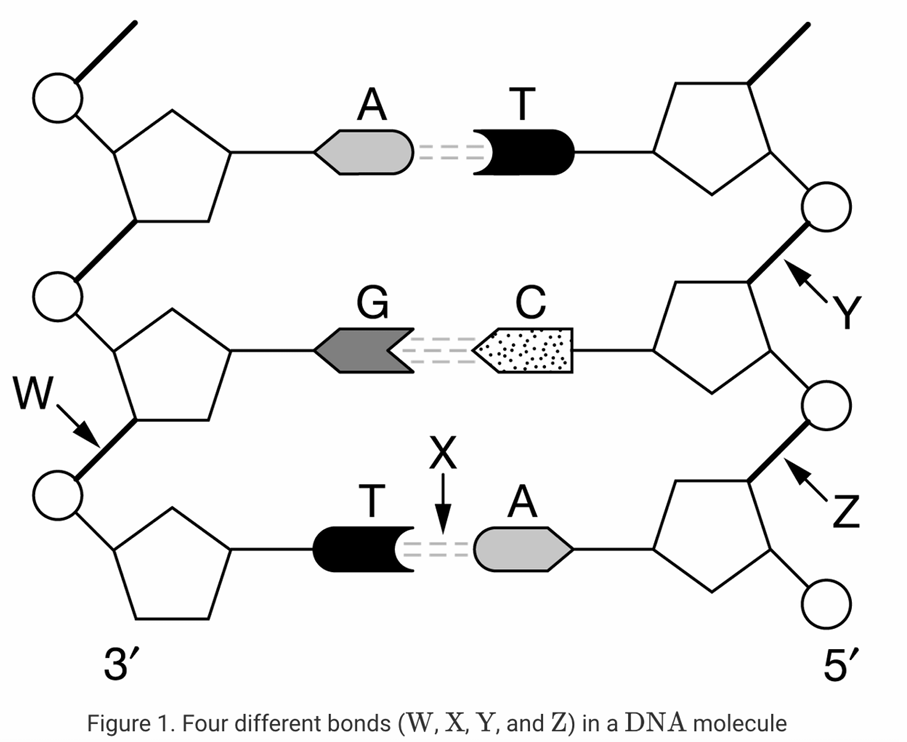
9. Which labeled bond(s) in the figure of a DNA molecule above are covalent bonds?
- A. Bond W only
- B. Bond X only
- C. Bonds Y and Z only
- D. Bonds W, Y, and Z only
10. Which labeled bond(s) in the figure of a DNA molecule above are hydrogen bonds?
- A. Bond W only
- B. Bond X only
- C. Bonds Y and Z only
- D. Bonds W, Y, and Z only
▶️ Answer/Explanation
9 Answer: D
The DNA molecule consists of a sugar-phosphate backbone and nitrogenous base pairs.
Labels W, Y, and Z point to the bonds connecting the phosphate groups and deoxyribose sugars.
These form the phosphodiester linkage, which acts as the structural backbone and consists of strong covalent bonds.
10 Answer: B
Label X points to the dashed lines connecting the complementary nitrogenous bases (Adenine and Thymine).
The two strands of the DNA helix are held together by these weak hydrogen bonds between the bases.
Therefore, only bond X represents a hydrogen bond.
Question 11

▶️ Answer/Explanation
Question 12 – 21
A. Cell wall
B. Mitochondria
C. Plasma membrane
D. Chloroplast
E. Nucleus
F. Endoplasmic reticulum
G. Ribosome
H. (Central) Vacuole
I. Lysosome
J. Golgi apparatus
K. Vesicle
▶️ Answer/Explanation
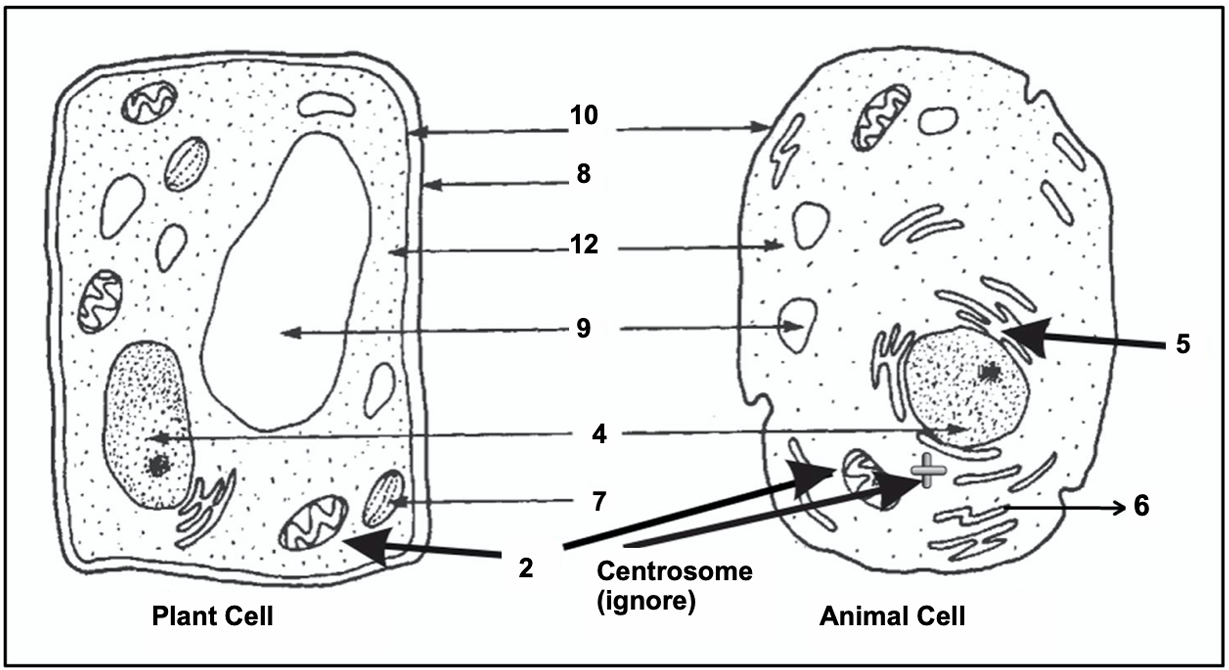
$12$. G. Ribosome
Ribosomes are the sites of protein synthesis.
They translate genetic information (mRNA) into polypeptide chains.
They can be found floating freely in the cytoplasm or attached to the rough Endoplasmic Reticulum.
$13$. B. Mitochondria
Mitochondria are known as the “powerhouse” of the cell.
They perform cellular respiration to generate $ATP$ (energy).
The inner membrane is folded into cristae to increase surface area for reactions.
$14$. I. Lysosome
Lysosomes are the cell’s waste disposal system.
They contain acidic hydrolytic enzymes.
These enzymes digest biomolecules, old organelles, and invading pathogens.
$15$. E. Nucleus
The nucleus houses the cell’s DNA (chromatin/chromosomes).
It regulates gene expression and cell division.
It is surrounded by a double membrane called the nuclear envelope.
$16$. F. Endoplasmic reticulum
The ER is a network of membranes involved in synthesis.
Rough ER (studded with ribosomes) produces and processes proteins.
Smooth ER synthesizes lipids and detoxifies chemicals.
$17$. J. Golgi apparatus
The Golgi apparatus acts as the cell’s “post office”.
It receives proteins from the ER, modifies them (e.g., glycosylation), and packages them into vesicles.
These vesicles are then transported to their specific destinations.
$18$. D. Chloroplast
Chloroplasts are found in plant cells and algae.
They contain chlorophyll which captures light energy.
This energy is used to convert $CO_2$ and water into glucose (photosynthesis).
$19$. A. Cell wall
The cell wall is a rigid outer layer found in plants, fungi, and bacteria.
In plants, it is composed mainly of cellulose.
It provides structural support and protection against mechanical stress.
$20$. H. (Central) Vacuole
Plant cells have a large central vacuole that takes up most of the cell’s volume.
It stores water, nutrients, and waste products.
The water pressure inside (turgor pressure) keeps the plant cell rigid and upright.
$21$. C. Plasma membrane
Also known as the cell membrane, it defines the cell boundary.
It is a phospholipid bilayer with embedded proteins (fluid mosaic model).
It is semi-permeable, regulating the transport of substances in and out of the cell.
Question 22
B. Membrane-bound organelles allow the cell to compartmentalize and specialize.
C. Membrane-bound organelles allow the cell to produce $ATP$ in any location.
D. Membrane-bound organelles allow the cell to evolve faster.
▶️ Answer/Explanation
The correct answer is B.
Membranes create distinct internal environments within the cell.
This compartmentalization allows incompatible chemical reactions to occur simultaneously.
Specific enzymes and substrates are concentrated in specialized areas, like the lysosome or mitochondria.
It increases metabolic efficiency by preventing interference between different pathways.
Other options like $ATP$ production (C) are restricted to specific organelles rather than “any location.”
Faster reproduction (A) and evolution (D) are not direct functions of internal membranes.
Question 23
B. lysosomes
C. ribosomes
D. chloroplasts
▶️ Answer/Explanation
The correct answer is not explicitly listed among the primary options, as lipid synthesis primarily occurs in the Smooth Endoplasmic Reticulum (SER).
However, looking at the provided choices:
Vacuoles are used for storage of water, nutrients, or waste.
Lysosomes contain digestive enzymes to break down macromolecules.
Ribosomes are the sites of protein synthesis, not lipids.
Chloroplasts are the site of photosynthesis and do synthesize some specific membrane lipids.
In most introductory biology contexts, if the Smooth ER is missing, the question may imply a defect in an organelle with metabolic capabilities like the chloroplast (in plants).
If this is a multiple-choice error, the Smooth ER is the standard biological answer for general lipid synthesis.
Question 24
B. It has pores that allow for molecules to move in and out of the nucleus.
C. It has a nucleolus region for the production of RNA.
D. The membrane is composed of tRNA.
▶️ Answer/Explanation
The correct answer is D.
The nuclear envelope is a double-membrane structure made of a phospholipid bilayer, not $\text{tRNA}$.
$\text{tRNA}$ (transfer RNA) is a molecule involved in protein synthesis, not a structural component of membranes.
The nucleus contains nuclear pores that regulate the trafficking of proteins and $\text{RNA}$.
The nucleolus is a dense region within the nucleus responsible for synthesizing ribosomal $\text{RNA}$ ($\text{rRNA}$).
Options A, B, and C all accurately describe anatomical features of the eukaryotic nucleus.
Question 25
B. Rough endoplasmic reticulum
C. Mitochondria
D. Vacuoles
▶️ Answer/Explanation
The correct answer is B. Rough endoplasmic reticulum.
Pancreas cells specialize in the synthesis and secretion of digestive enzymes.
Since enzymes are proteins, they are synthesized by ribosomes.
The Rough Endoplasmic Reticulum (RER) is studded with ribosomes and is the primary site for protein synthesis.
Cells active in secretion require an extensive $RER$ network to process these proteins.
In contrast, the Smooth ER is primarily involved in lipid synthesis and detoxification.
Mitochondria provide energy ($ATP$), and Vacuoles are used for storage, making them less central to the specific task of enzyme production.
Question 26

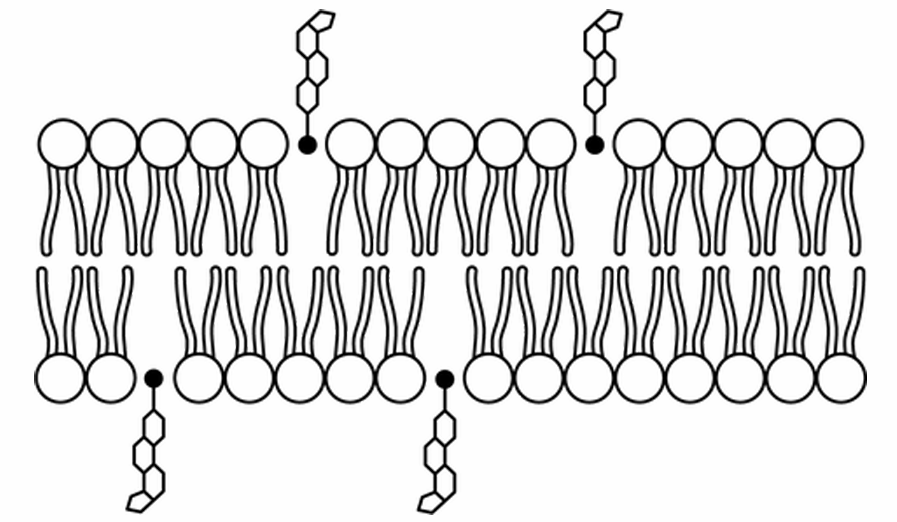
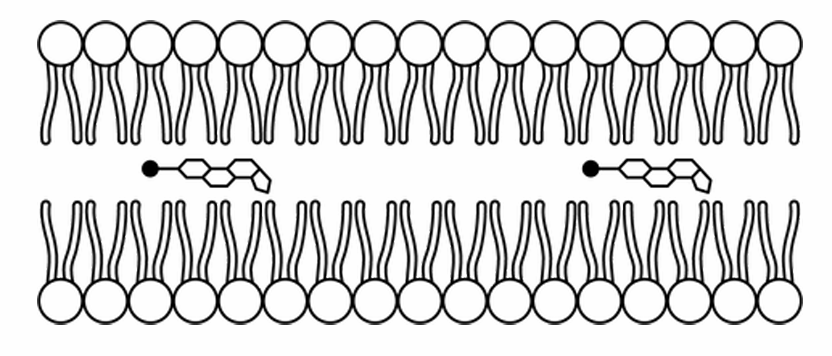
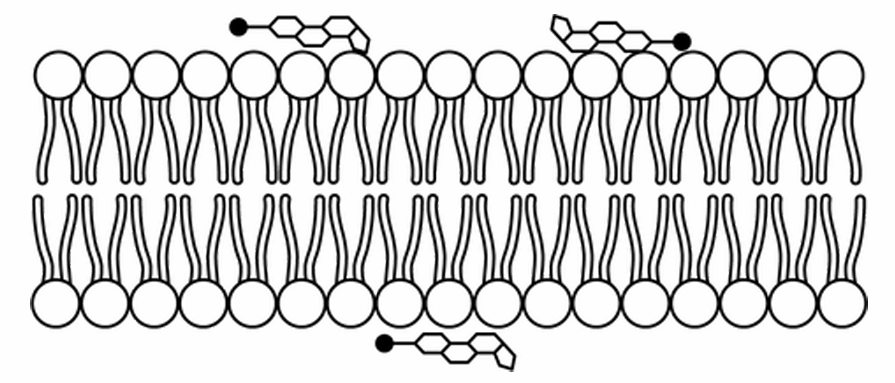
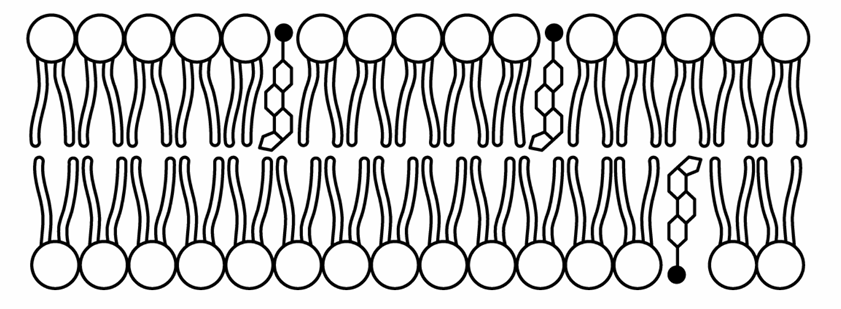
▶️ Answer/Explanation
The correct option is D.
Phospholipids are amphipathic, containing hydrophilic phosphate heads and hydrophobic fatty acid tails.
Cholesterol is also amphipathic, possessing a small polar hydroxyl head and a large nonpolar hydrocarbon body.
Thermodynamically, the polar head of cholesterol must align with the polar heads of the phospholipids to interact with water.
The nonpolar region of cholesterol inserts itself between the hydrophobic tails of the bilayer.
Model D correctly depicts the cholesterol heads pointing toward the aqueous environment, integrated into the surface.
This orientation allows cholesterol to regulate membrane fluidity by preventing tight packing of tails.
Question 27
▶️ Answer/Explanation
The correct option is C.
Enzymes are proteins, and secretory proteins are synthesized by ribosomes on the Rough ER.
These proteins are then transported via vesicles to the next destination.
The Golgi apparatus modifies, sorts, and packages these proteins for secretion.
Since the pancreas is a secretory organ, it requires a highly developed endomembrane system.
Options A and B are incorrect as they include chloroplasts, which are only found in plant cells.
Option D is less likely because lysosomes are for intracellular digestion, not extracellular secretion.
Question 28
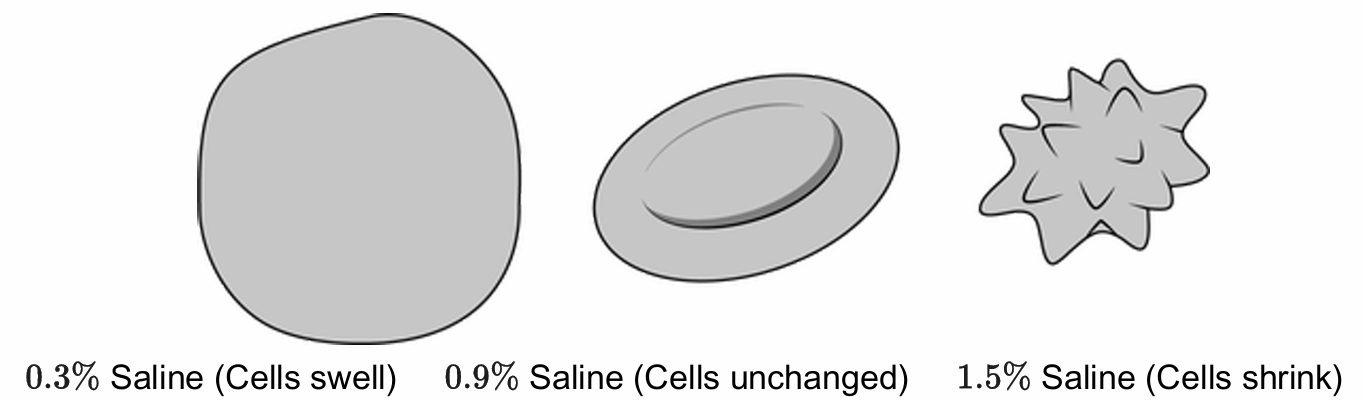
B. The cell placed in the $0.9\%$ saline solution.
C. The cell placed in the $1.5\%$ saline solution.
D. This cannot be determined from the information provided.
▶️ Answer/Explanation
Correct Option: A
A hypotonic solution has a lower solute concentration than the inside of the cell.
This causes water to enter the cell via osmosis to reach equilibrium.
The net movement of water into the cell causes it to swell or potentially burst.
The diagram shows the cells swell in the $0.3\%$ saline solution.
In contrast, the $0.9\%$ solution is isotonic (no change) and $1.5\%$ is hypertonic (shrinkage).
Therefore, the $0.3\%$ saline solution represents the hypotonic condition.
Question 29
B. By facilitated diffusion
C. By active transport
D. By endocytosis
▶️ Answer/Explanation
The plasma membrane consists of a hydrophobic phospholipid bilayer.
Aldosterone is described as a small, nonpolar, and hydrophobic molecule.
Hydrophobic molecules can dissolve in and pass directly through the lipid bilayer.
The movement occurs down a concentration gradient, indicating passive transport.
Since no protein channel or carrier is mentioned for this specific passage, it is simple diffusion.
Facilitated diffusion and active transport are ruled out as they require specialized proteins.
Endocytosis is unnecessary for small, membrane-permeable molecules like steroid hormones.
Question 30
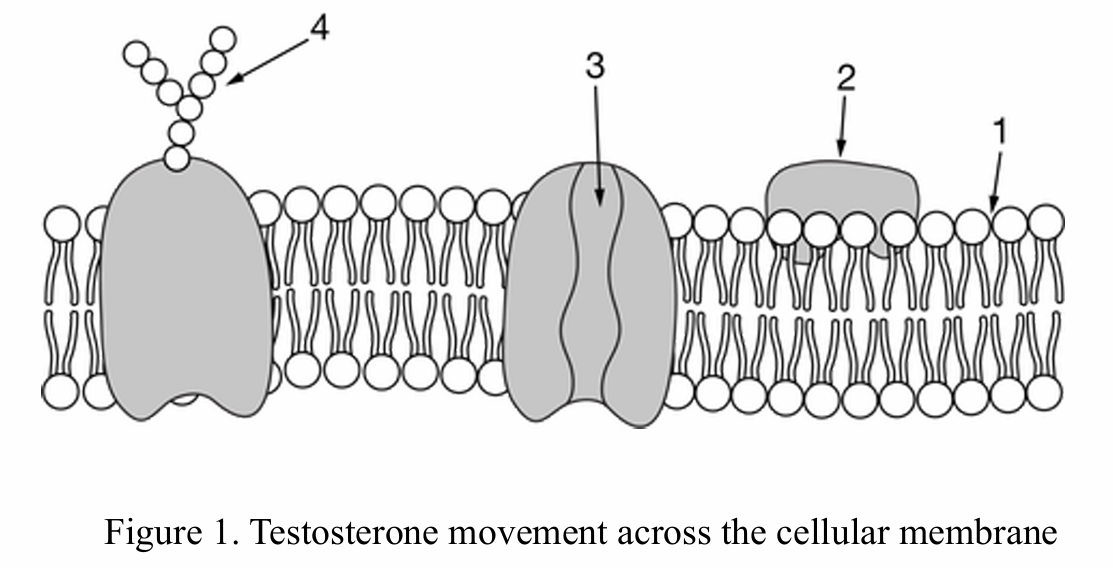
B. $2$, because oxygen covalently binds to a surface protein and transports into the cell.
C. $3$, because oxygen must dissolve in water to flow through a protein channel across the membrane.
D. $4$, because oxygen is taken into the cell by endocytosis after binding to a glycoprotein.
▶️ Answer/Explanation
The correct answer is A.
Label $1$ represents the phospholipid bilayer.
Oxygen ($O_2$) is a small, nonpolar molecule.
Nonpolar molecules can dissolve in the hydrophobic lipid core.
This allows $O_2$ to cross the membrane via simple diffusion.
It does not require transport proteins ($2$ or $3$) or carbohydrates ($4$).
Movement occurs down the concentration gradient without energy input.
Question 31
B. 2, because $Ca^{2+}$ ions will covalently binds to a surface protein to be transported into the cell.
C. 3, because $Ca^{2+}$ ions must flow through a protein channel across the membrane because they are charged.
D. 4, because $Ca^{2+}$ ions are taken into the cell by endocytosis after binding to a glycoprotein.
▶️ Answer/Explanation
Correct Option: C
$Ca^{2+}$ ions are hydrophilic and highly charged, preventing them from passing through the hydrophobic lipid bilayer.
These ions require facilitated diffusion via specific transmembrane protein channels (represented by point 3).
Simple diffusion (Option A) is impossible because ions are polar/charged, not nonpolar.
Covalent bonding to surface proteins (Option B) is not a standard mechanism for ion transport.
The protein channel provides a hydrophilic pathway that bypasses the fatty acid tails of the phospholipids.
This allows the cell to regulate the internal concentration of $Ca^{2+}$ effectively.
Question 32 – 34
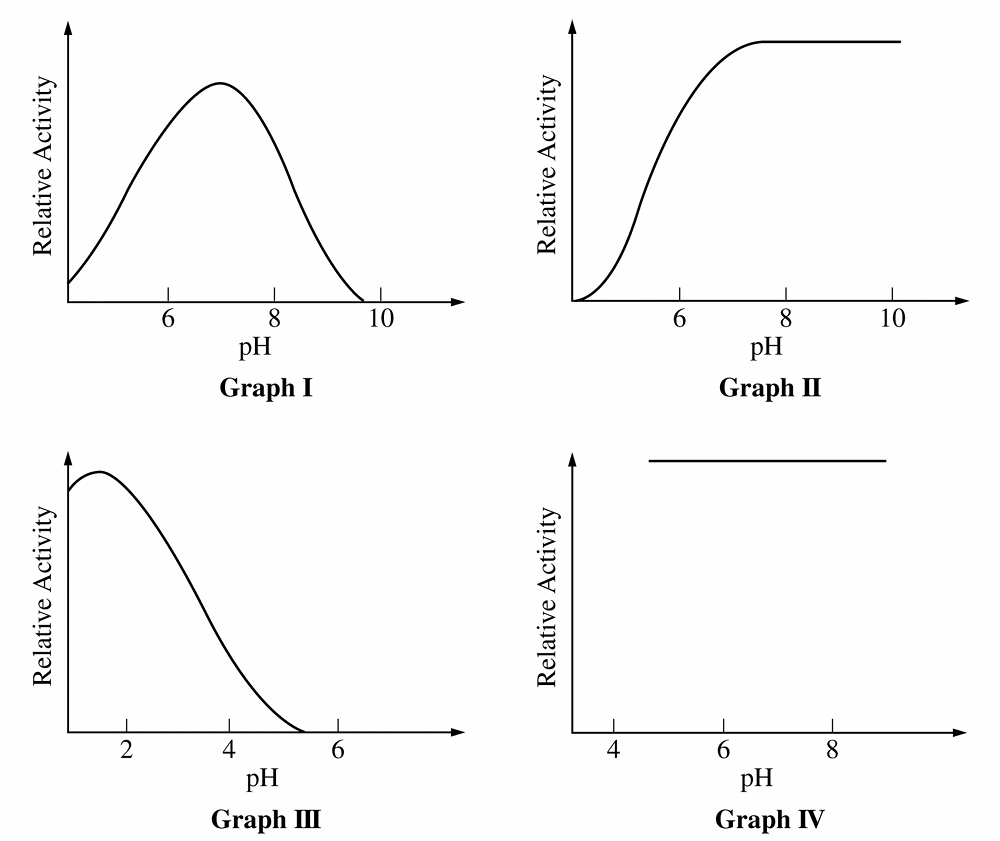
B. \(II\) only
C. \(III\) only
D. \(I\) and \(III\) only
B. \(IV\) only
C. \(II\) and \(III\) only
D. \(I\), \(II\), and \(III\) only
B. \(pH\) affects the temperature of the reaction
C. the enzyme has a quaternary structure
D. the enzyme has disulfide bonds
▶️ Answer/Explanation
33. Answer: D (I, II, and III only)
34. Answer: A
For Q32, Graph \(III\) peaks at acidic \(pH\) (\(\approx 2\)) and Graph \(I\) peaks at neutral \(pH\) (\(\approx 7\)).
Graph \(II\) shows activity increasing and remaining high in the alkaline range (\(pH\) \(8\)-\(10\)), identifying it as the alkaline enzyme.
For Q33, “sensitive” means enzyme activity varies as \(pH\) changes.
Graph \(IV\) is a horizontal line, indicating activity is constant and independent of \(pH\) (insensitive).
Graphs \(I\), \(II\), and \(III\) all show curves where activity changes with \(pH\), making them the correct choice.
For Q34, enzyme activity depends on the precise shape of the active site.
Changes in \(pH\) alter the charge of amino acid side chains, disrupting the bonds holding the active site’s shape.
This loss of specific shape prevents substrate binding, explaining the decline in activity seen in Graph \(I\).
Question 35 – 38
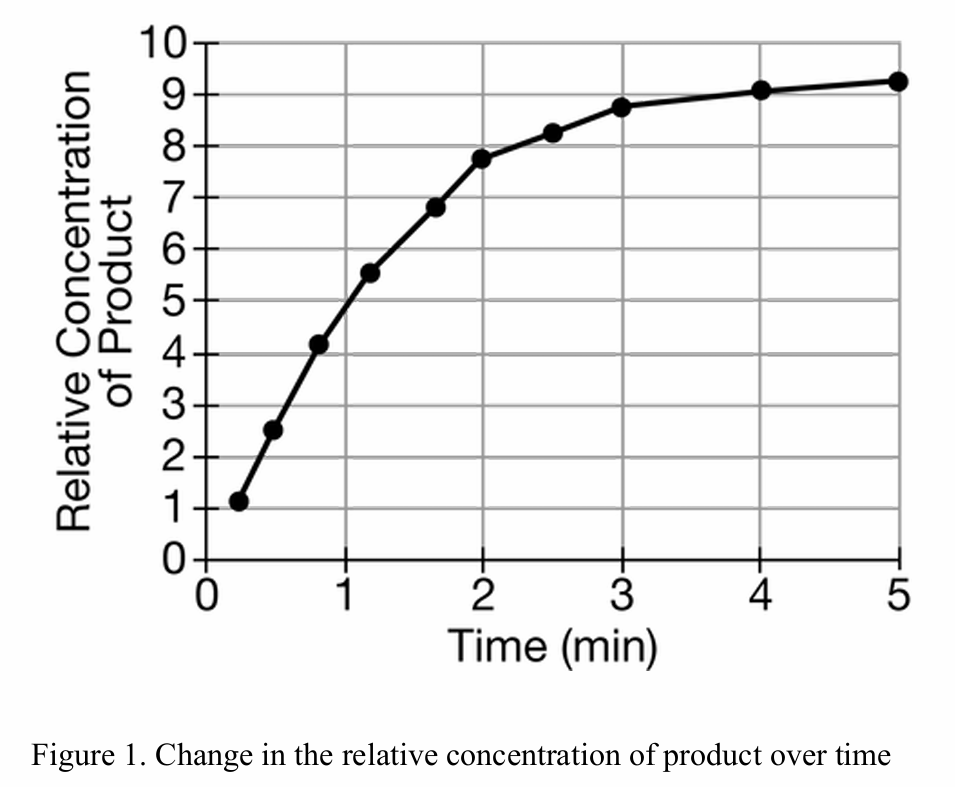
35. How is this rate of this enzymatic chemical reaction being measured? (Hint: What is the DV?)
B. The time.
C. Substrates consumed.
D. The change in pH.
36. Which of the following is another method to measure the rate of an enzyme catalyzed reaction?
B. The rate that substrates/reactants are consumed.
C. The change in pH over time.
D. The change in temperature over time.
37. Which of the following best explains how the rate of the reaction changes over time?
B. The rate increases because the ratio of product to substrate increases.
C. The rate decreases because the ratio of product to substrate increases.
D. The rate decreases because the enzyme is used up as the reaction progresses.
38. If a competitive inhibitor were added at one minute time for this reaction, predict the relative concentration of product produced at time 2 minutes?
B. Between \(3\) and \(5\).
C. Between \(5\) and \(7\).
D. Between \(8\) and \(10\).
▶️ Answer/Explanation
35. Correct Answer: A
The dependent variable (DV) is the variable being measured and is typically plotted on the Y-axis. The Y-axis label in Figure 1 is “Relative Concentration of Product”. Therefore, the rate is being measured by tracking the relative product produced over time.
36. Correct Answer: B
The rate of an enzymatic reaction can be determined by measuring the appearance of product or the disappearance of substrate. Since the conversion of reactants to products is stoichiometric, measuring the rate at which substrates/reactants are consumed is a valid alternative method.
37. Correct Answer: C
The slope of the curve (representing the rate) flattens over time, indicating the rate is decreasing. This happens because the substrate concentration drops as it is converted to product. Option D is incorrect because enzymes are not consumed in the reaction. Option C correctly describes the kinetic shift: as product accumulates and substrate depletes (increasing the product:substrate ratio), the reaction slows.
38. Correct Answer: C
At \(t=1\) minute, the product concentration is approximately \(4.2\). Under normal conditions, at \(t=2\) minutes, it reaches approximately \(7.8\). A competitive inhibitor would slow the reaction rate but not stop it immediately. Therefore, the product concentration would increase from \(4.2\), but to a level lower than the uninhibited \(7.8\). The range of \(5\) to \(7\) fits a reaction that is proceeding at a reduced rate.
Question 39
B. During the light reactions of photosynthesis, water is split, removing electrons and protons, and oxygen gas is released.
C. During the Calvin cycle, water is split, regenerating $\text{NADPH}$ from $\text{NADP}^+$, and oxygen gas is released.
D. During the Calvin cycle, water is split, the hydrogen atoms are added to intermediates of sugar synthesis, and oxygen gas is released.
▶️ Answer/Explanation
During the light-dependent reactions, light energy drives the photolysis (splitting) of water molecules.
This process occurs at Photosystem II to replace electrons lost to the electron transport chain.
Water molecules are broken down into protons ($H^+$), electrons ($e^-$), and oxygen gas ($O_2$).
Since the oxygen atoms in the reactant $\text{H}_2\text{O}$ are labeled with $^{18}\text{O}$, the resulting $\text{O}_2$ gas carries that label.
The Calvin cycle (Options C and D) does not involve the splitting of water or the release of oxygen.
Hydrogen from water does not combine directly with $\text{CO}_2$ (Option A); it is carried by $\text{NADPH}$ to the Calvin cycle.
Question 40
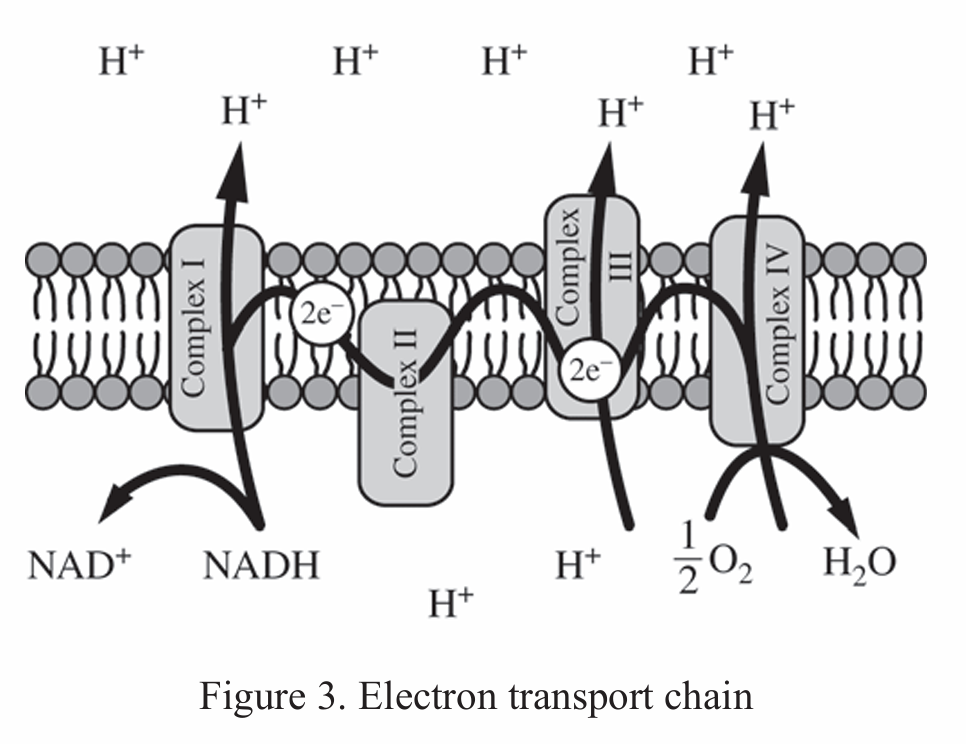
B. This ETC is located in the inner membrane of the mitochondria, because it uses $\text{NADH}$ as an electron carrier and oxygen as the final electron acceptor.
C. This ETC is located in the thylakoid membrane of chloroplasts, because it contains Photosystems and uses water as a reactant.
D. This ETC is located in the inner membrane of the mitochondria, because it generates an $\text{H}^+$ gradient.
▶️ Answer/Explanation
The diagram shows $\text{NADH}$ being oxidized to $\text{NAD}^+$ at Complex I.
It illustrates electrons moving through complexes to reduce $\frac{1}{2} \text{O}_2$ into $\text{H}_2\text{O}$.
In chloroplasts, the final electron acceptor is $\text{NADP}^+$, not oxygen.
The presence of $\text{NADH}$ as the primary donor is characteristic of mitochondrial respiration.
While both types of ETCs create an $\text{H}^+$ gradient, only B provides a specific, correct identifier.
Therefore, the correct location is the inner mitochondrial membrane.
Correct Option: B
Question 41
B. The ETC splits water to make oxygen gas, and ATP synthase phosphorylates oxygen to make $CO_2$.
C. The ETC creates NADH requires oxygen gas, and ATP synthase phosphorylates oxygen to make $CO_2$.
D. The ETC splits water to make oxygen, electrons, and $H^+$, and ATP synthase phosphorylates ADP to make ATP.
▶️ Answer/Explanation
The correct option is A.
In the Electron Transport Chain (ETC), electrons are passed along proteins to oxygen, the final electron acceptor.
This movement provides energy to pump $H^+$ ions across the membrane, creating an electrochemical gradient.
Oxygen gas ($O_2$) is essential here to accept electrons and form water ($H_2O$).
ATP synthase then uses the flow of $H^+$ ions back across the membrane to power the phosphorylation of ADP.
This process adds a phosphate group to $ADP$ to synthesize $ATP$.
Options B, C, and D are incorrect as they misidentify the substrates or the role of water and $CO_2$.
Question 42
▶️ Answer/Explanation
The correct option is C.
The chloroplast is divided into the thylakoids and the stroma.
Light-dependent reactions (Options A, B, and D) occur within or across the thylakoid membranes.
The Calvin cycle (light-independent reactions) takes place specifically in the aqueous stroma.
Carbon fixation is the first step of the Calvin cycle, where \(\text{CO}_2\) is attached to \(\text{RuBP}\).
This process utilizes the \(\text{ATP}\) and \(\text{NADPH}\) produced by the light reactions.
Therefore, carbon fixation is the only process listed that occurs in the stroma.
Question 43
B. water
C. carbon dioxide
D. NADPH
▶️ Answer/Explanation
The correct answer is B. water.
During the light-dependent reactions of photosynthesis, light energy is absorbed by chlorophyll.
This energy is used to split molecules of $\text{H}_2\text{O}$ in a process called photolysis.
The reaction can be represented as: $2\text{H}_2\text{O} \rightarrow 4\text{H}^+ + 4e^- + \text{O}_2$.
The electrons ($e^-$) and protons ($\text{H}^+$) are used to generate $\text{ATP}$ and $\text{NADPH}$.
The oxygen atoms from the water molecules combine to form oxygen gas ($\text{O}_2$).
This $\text{O}_2$ is then released as a byproduct into the atmosphere.
Carbon dioxide ($\text{CO}_2$) is used later in the Calvin cycle to build glucose, not to produce oxygen.
Question 44
B. ATP synthase
C. ETC
D. Calvin cycle
▶️ Answer/Explanation
The correct answer is A. Photosystems.
Chlorophylls and carotenoids are embedded within the thylakoid membranes.
These pigments are organized into functional units called photosystems (PS I and PS II).
Photosystems harvest light energy to excite electrons during the light-dependent reactions.
While the ETC and ATP synthase are also in the membrane, they do not house the pigments.
The Calvin cycle occurs in the stroma and does not contain light-absorbing pigments.
Question 45
B. $\text{ATP}$
C. $\text{NADPH}$
D. Sugars such as glucose
▶️ Answer/Explanation
The correct answer is D. Sugars such as glucose.
Light-dependent reactions occur in the thylakoid membranes of the chloroplast.
Chlorophyll absorbs solar energy to split water molecules, releasing $\text{O}_2$ as a byproduct.
This process generates chemical energy in the form of $\text{ATP}$ via photophosphorylation.
It also produces reducing power in the form of $\text{NADPH}$ for later use.
Sugars are synthesized during the light-independent reactions (Calvin Cycle).
The Calvin Cycle uses the $\text{ATP}$ and $\text{NADPH}$ produced earlier to fix $\text{CO}_2$.
Question 46
B. Water
C. $\text{RuBP}$
D. $\text{NADPH}$
▶️ Answer/Explanation
The correct answer is A. Carbon dioxide.
In the Calvin cycle, $\text{CO}_2$ molecules enter the chloroplast from the atmosphere.
The enzyme Rubisco fixes inorganic carbon from $\text{CO}_2$ into an organic molecule.
This process is known as carbon fixation, the first step of sugar synthesis.
While $\text{RuBP}$ acts as the acceptor, the actual “building block” atoms come from $\text{CO}_2$.
$\text{ATP}$ and $\text{NADPH}$ provide the energy and electrons, but no carbon.
Ultimately, these fixed carbon atoms are rearranged to form $\text{G3P}$ and glucose.
Question 47
▶️ Answer/Explanation
The correct answer is C. $\text{O}_2$.
The Calvin cycle is the light-independent phase of photosynthesis occurring in the stroma.
It requires $\text{CO}_2$ as the carbon source for fixation.
It utilizes $\text{ATP}$ and $\text{NADPH}$ produced during the light-dependent reactions.
$\text{O}_2$ is actually a byproduct of the light reactions (photolysis of water).
In fact, high levels of $\text{O}_2$ can inhibit the cycle through photorespiration.
Question 48
B. Electrons are fused together with $ADP$ to form $ATP$.
C. Glucose is produced by $ATP$ synthase.
D. Carbon dioxide is produced via dehydration synthesis.
▶️ Answer/Explanation
The correct option is A.
When photons of light strike the antenna pigments of Photosystem II ($PSII$), the energy is funneled to the reaction center ($P680$).
This energy boosts electrons to a higher energy level, causing them to be captured by a primary electron acceptor.
These excited electrons are then passed into the electron transport chain ($ETC$) to create a proton gradient.
The “hole” left in $PSII$ is filled by electrons obtained from the photolysis of $H_2O$.
Options B and C are incorrect as $ATP$ and glucose are products of later stages or the Calvin cycle.
Option D is incorrect because $CO_2$ is consumed in photosynthesis, not produced via dehydration synthesis.
Question 49
B. Not having enough pigments to produce $\text{NADPH}$ via the $\text{ETC}$.
C. Not having enough electrons to excite (become high energy) for the Photosystems and the $\text{ETC}$.
D. Not having enough oxygen to produce $\text{ATP}$ in the light-independent reactions.
▶️ Answer/Explanation
The correct option is C.
In the light-dependent reactions, water molecules ($\text{H}_2\text{O}$) undergo photolysis.
This process splits water to provide electrons ($e^-$) to Photosystem II ($\text{PSII}$).
These electrons replace those lost by chlorophyll when they are excited by sunlight.
Without sufficient water, there is a shortage of electrons to enter the Electron Transport Chain ($\text{ETC}$).
This halts the production of energy carriers like $\text{ATP}$ and $\text{NADPH}$.
Ultimately, the lack of high-energy electrons stops the entire photosynthetic process.
Question 50
B. A second messenger molecule is synthesized by a kinase.
C. A ligand binds to a receptor, activating it and changing its shape.
D. Specific proteins are synthesized and phosphorylated.
▶️ Answer/Explanation
The correct answer is C.
Cell signaling begins when a ligand (chemical messenger) recognizes and binds to a specific receptor.
This binding event causes a conformational change (shape change) in the receptor protein.
This structural shift is the initial trigger that initiates the signal transduction pathway.
Options A, B, and D describe downstream events that occur after the initial reception.
Reception must occur at the cell surface or cytosol before transduction or response can begin.
