IB DP Chemistry - Structure 2.4 From models to materials - IB Style Questions For HL Paper 1A -FA 2025
Question
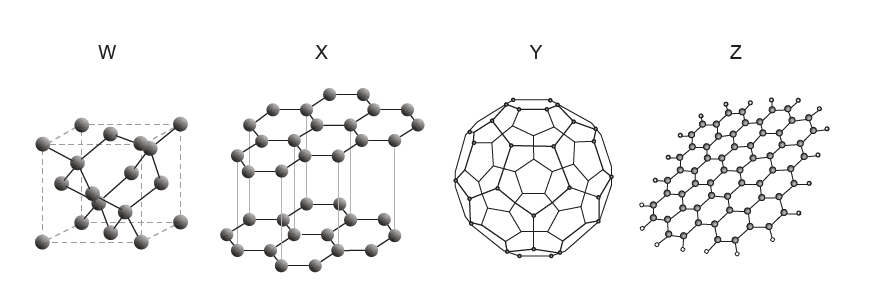
| Option | Graphene | Fullerene | Graphite | Diamond |
|---|---|---|---|---|
| A | Z | Y | X | W |
| B | W | Z | Y | X |
| C | X | W | Z | Y |
| D | Y | X | W | Z |
▶️ Answer/Explanation
Correct Answer: A
• Z shows a single sheet of hexagons → Graphene.
• Y is a spherical molecule → Fullerene.
• X has stacked layers → Graphite.
• W is a tetrahedral network → Diamond.
Question
What are the hybridizations of the atoms labelled 1, 2 and 3 in the following molecule?
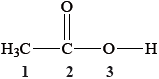
| Option | 1 | 2 | 3 |
|---|---|---|---|
| A | sp2 | sp2 | sp |
| B | sp3 | sp2 | sp3 |
| C | sp2 | sp | sp3 |
| D | sp3 | sp2 | sp |
▶️ Answer/Explanation
Correct option: B
• Atom 1 (CH3 carbon) forms four σ bonds ⇒ hybridization = sp3.
• Atom 2 (carbonyl carbon) forms three σ bonds and one π bond ⇒ trigonal planar ⇒ sp2.
• Atom 3 (–O–H oxygen) has two lone pairs and two σ bonds ⇒ four electron domains ⇒ sp3.
Question
II. Graphite
III. \(C_{60}\) fullerene
B. I and III only
C. II and III only
D. I, II and III
▶️ Answer/Explanation
Diamond has a tetrahedral structure where each carbon forms four single bonds → this is \(sp^3\) hybridization. Therefore, diamond is not \(sp^2\)-hybridized.
Graphite has layers of carbon atoms arranged in hexagonal rings. Each carbon is bonded to three others → this is classic \(sp^2\) hybridization.
\(C_{60}\) fullerene (buckminsterfullerene) also has carbon atoms bonded to three neighbors in a spherical structure → this is also \(sp^2\) hybridization.
Therefore, the allotropes showing \(sp^2\) hybridization are:
• Graphite
• \(C_{60}\) fullerene
✅ Answer: (C)
