IB PHYSICS HL(Higher level) – 2024 – Practice Questions- All Topics
Topic 12.1 The interaction of matter wih radiation
Topic 12 Weightage : 7 %
All Questions for Topic 12.1 – Photons , The photoelectric effect , Matter waves , Pair production and pair annihilation , Quantization of angular momentum in the Bohr model for hydrogen , The wave function , The uncertainty principle for energy and time and position and momentum , Tunnelling, potential barrier and factors affecting tunnelling probability
Yellow light of photon energy 3.5 x 10–19 J is incident on the surface of a particular photocell.
The photocell is connected to a cell as shown. The photoelectric current is at its maximum value (the saturation current).
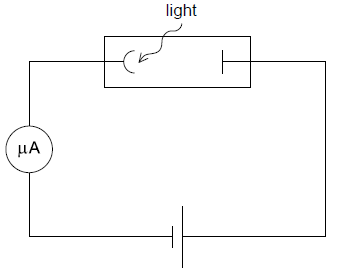
Radiation with a greater photon energy than that in (b) is now incident on the photocell. The intensity of this radiation is the same as that in (b).
a.i. Calculate the wavelength of the light. [1]
▶️Answer/Explanation
Markscheme
a.i.
wavelength = «\(\frac{{hc}}{E} = \frac{{1.99 \times {{10}^{ – 25}}}}{{{\text{3}}{\text{.5}} \times {\text{1}}{{\text{0}}^{ – 19}}}} = \)» 5.7 x 10–7 «m»
If no unit assume m.
«potential» energy is required to leave surface
Do not allow reference to “binding energy”.
Ignore statements of conservation of energy.
all/most energy given to potential «so none left for kinetic energy»
energy surplus = 1.7 x 10–19 J
vmax = \(\sqrt {\frac{{2 \times 1.7 \times {{10}^{ – 19}}}}{{9.1 \times {{10}^{ – 31}}}}} = 6.1 \times {10^5}\) «m s–1»
Award [1 max] if surplus of 5.2 x 10–19J used (answer: 1.1 x 106 m s–1)
«same intensity of radiation so same total energy delivered per square metre per second»
light has higher photon energy so fewer photons incident per second
Reason is required
1:1 correspondence between photon and electron
so fewer electrons per second
current smaller
Allow ECF from (c)(i)
Allow ECF from MP2 to MP3.
Question
The de Broglie wavelength λ of a particle accelerated close to the speed of light is approximately
λ =
where E is the energy of the particle.
A beam of electrons of energy 4.2 × 108 eV is produced in an accelerator.
(a) Show that the wavelength of an electron in the beam is about 3 × 10–15 m. [1]
▶️Answer/Explanation
Ans
\(\lambda =\frac{6.63\times10^{-34}\times3\times10^8}{1.60\times10^{-19}\times4.2\times10^8} = 2.96\times10^{-15}m\)
Hydrogen atoms in an ultraviolet (UV) lamp make transitions from the first excited state to the ground state. Photons are emitted and are incident on a photoelectric surface as shown.
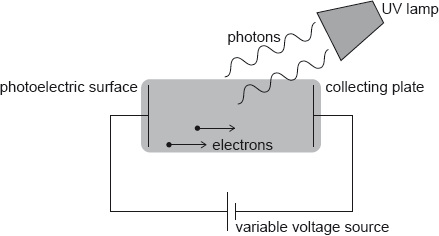
The photons cause the emission of electrons from the photoelectric surface. The work function of the photoelectric surface is 5.1 eV.
The electric potential of the photoelectric surface is 0 V. The variable voltage is adjusted so that the collecting plate is at –1.2 V.

a. Show that the energy of photons from the UV lamp is about 10 eV. [2]
▶️Answer/Explanation
Markscheme
a.
E1 = –13.6 «eV» E2 = – \(\frac{{13.6}}{4}\) = –3.4 «eV»
energy of photon is difference E2 – E1 = 10.2 «≈ 10 eV»
Must see at least 10.2 eV.
[2 marks]
10 – 5.1 = 4.9 «eV»
4.9 × 1.6 × 10–19 = 7.8 × 10–19 «J»
Allow 5.1 if 10.2 is used to give 8.2×10−19 «J».
EPE produced by battery
exceeds maximum KE of electrons / electrons don’t have enough KE
For first mark, accept explanation in terms of electric potential energy difference of electrons between surface and plate.
[2 marks]
4.9 «V»
Allow 5.1 if 10.2 is used in (b)(i).
Ignore sign on answer.
[1 mark]
two equally spaced vertical lines (judge by eye) at approximately 1/3 and 2/3
labelled correctly
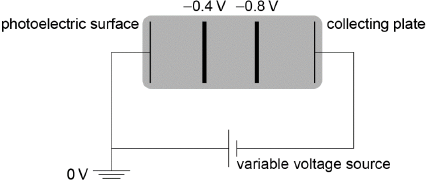
[2 marks]
kinetic energy at collecting plate = 0.9 «eV»
speed = «\(\sqrt {\frac{{2 \times 0.9 \times 1.6 \times {{10}^{ – 19}}}}{{9.11 \times {{10}^{ – 31}}}}} \)» = 5.6 × 105 «ms–1»
Allow ECF from MP1
[2 marks]
Yellow light of photon energy 3.5 x 10–19 J is incident on the surface of a particular photocell.

The photocell is connected to a cell as shown. The photoelectric current is at its maximum value (the saturation current).

Radiation with a greater photon energy than that in (b) is now incident on the photocell. The intensity of this radiation is the same as that in (b).
a.i. Calculate the wavelength of the light. [1]
▶️Answer/Explanation
Markscheme
a.i.
wavelength = «\(\frac{{hc}}{E} = \frac{{1.99 \times {{10}^{ – 25}}}}{{{\text{3}}{\text{.5}} \times {\text{1}}{{\text{0}}^{ – 19}}}} = \)» 5.7 x 10–7 «m»
If no unit assume m.
«potential» energy is required to leave surface
Do not allow reference to “binding energy”.
Ignore statements of conservation of energy.
all/most energy given to potential «so none left for kinetic energy»
energy surplus = 1.7 x 10–19 J
vmax = \(\sqrt {\frac{{2 \times 1.7 \times {{10}^{ – 19}}}}{{9.1 \times {{10}^{ – 31}}}}} = 6.1 \times {10^5}\) «m s–1»
Award [1 max] if surplus of 5.2 x 10–19J used (answer: 1.1 x 106 m s–1)
«same intensity of radiation so same total energy delivered per square metre per second»
light has higher photon energy so fewer photons incident per second
Reason is required
1:1 correspondence between photon and electron
so fewer electrons per second
current smaller
Allow ECF from (c)(i)
Allow ECF from MP2 to MP3.
