IB PHYSICS HL(Higher level) – 2024 – Practice Questions- All Topics
Topic 7.3 The structure of matter
Topic 7 Weightage : 3 %
All Questions for Topic 7.3 – Quarks, leptons and their antiparticles , Hadrons, baryons and mesons , The conservation laws of charge, baryon number, lepton number and strangeness , The nature and range of the strong nuclear force, weak nuclear force and electromagnetic force , Exchange particles , Feynman diagrams , Confinement , The Higgs boson
Question
Three conservation laws in nuclear reactions are
conservation of charge
conservation of baryon number
conservation of lepton number.
The reaction
n → π– + e++ \(\bar{v}_e\)
is proposed.
Which conservation laws are violated in the proposed reaction?
I and II only
I and III only
II and III only
I, II and III
▶️Answer/Explanation
Ans: C
\(\;\;\;\;n\rightarrow \pi^-+e^++\bar{\nu}_e\)
\(Q :0 = -1 + 1\) Conserved
\(B :1 = 0 + 0 +0\) Not Conserved
\(L :0 = 0 – 1 -1\) Not Conserved
Question
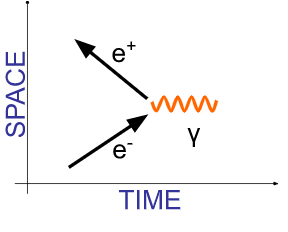
Which Feynman diagram describes the annihilation of an electron and its antiparticle?
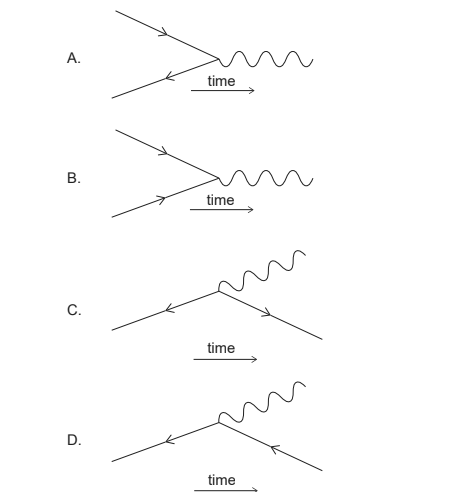
▶️Answer/Explanation
Ans: A
Question
In a hydrogen atom, the sum of the masses of a proton and of an electron is larger than the mass of the atom. Which interaction is mainly responsible for this difference?
A Electromagnetic
B Strong nuclear
C Weak nuclear
D Gravitational
▶️Answer/Explanation
Ans: A
Particles can be classified in term of the interactions they take part in:
The electron and proton have charge so take part in electromagnetic interactions.
The proton and neutron are nucleons so take part in nuclear interactions.
The neutrino only takes part in weak interactions (beta decay).
Question
Identify the conservation law violated in the proposed reaction.
p+ + p+ → p+ + n0 + μ+
A. Strangeness
B. Lepton number
C. Charge
D. Baryon number
▶️Answer/Explanation
Markscheme
B
\(\;\;\;\;n\rightarrow \pi^-+e^++\bar{\nu}_e\)
\(Q :1+1 = -1 +0+ 1\) Charge Conserved
\(L_\mu :0+0 = 0 + 0 -1\) Lepton Number Not Conserved
\(B :1+1 = 1 +1 +0\) Baryon Number Conserved
\(S :0+0 = 0 +0 +0\) Strangeness Number Conserved
Question
What is correct about the Higgs Boson?
A. It was predicted before it was observed.
B. It was difficult to detect because it is charged.
C. It is not part of the Standard Model.
D. It was difficult to detect because it has no mass.
▶️Answer/Explanation
Markscheme
A
The Higgs boson
Although we haven’t done all the mathematics, the particles of the standard model are all predicted by quantum mechanics. However, there is one piece that doesn’t fit. According to these predictions the W and Z bosons should not have any mass, but to fit in with the observation that the interactions are short range they must have large masses. One solution to this problem would be if the mass was not a property of the particle but a property of space. This is known as the Higgs field, and if there is a field there should be a particle associated with it, which we call the Higgs boson. A particle with these properties was detected in the ATLAS and CMS experiments at CERN on 4 July 2012.
Question
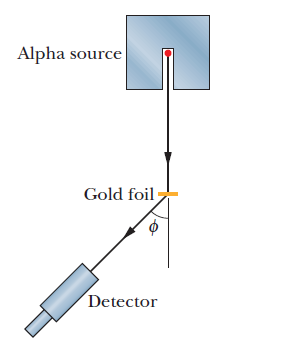
The structure of the atom was investigated by firing alpha particles from a source at a thin foil of gold. The basic set-up of the apparatus is shown.
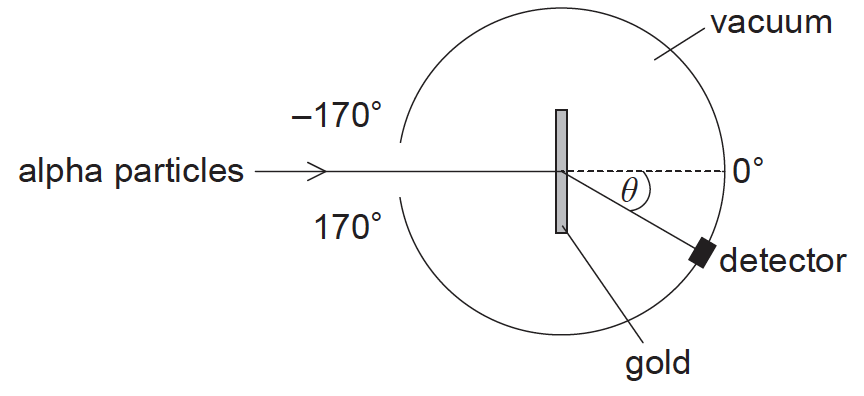
Which graph shows the variation in the number of scattered alpha particles with scattering angle \(\theta \)?
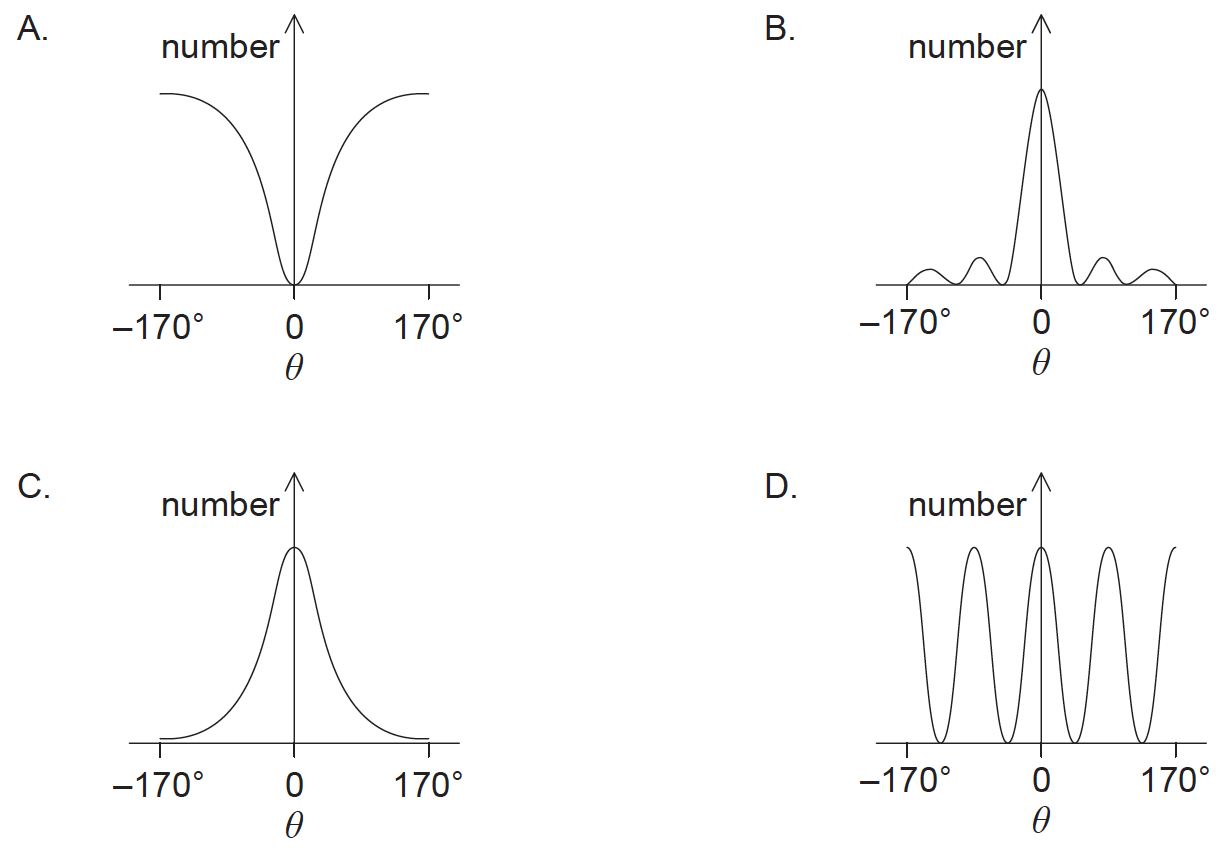
▶️Answer/Explanation
Markscheme
C
Rutherford derived a theoretical formula for the scattering of alpha particles from nuclei.
\(Nsin^4(\frac{\theta}{2}) =constant\)
where N is the number of scattered alpha particles. As the alpha particles approach the nucleus at higher energies deviations from the Rutherford model are observed due to the nuclear force.
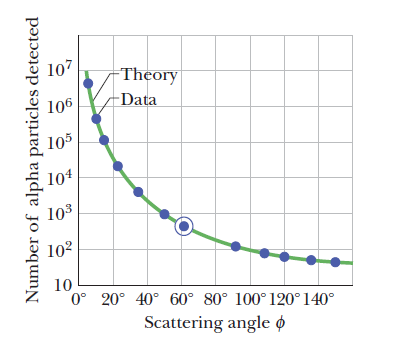
The dots are alpha-particle scattering data for a gold foil, obtained by Geiger and Marsden using the apparatus of Fig. 1.The solid curve is the theoretical prediction, based on the assumption that the atom has a small, massive, positively charged nucleus.The data have been adjusted to fit the theoretical curve at the experimental point that is enclosed in a circle.
Question
An alpha particle is directed head-on towards a nucleus of an isotope of iron. A second alpha particle, with the same energy as the first, is directed head-on towards a different isotope of iron.
Which of the following is a comparison of the distances of closest approach of the two alpha particles and the forces experienced by the alpha particles at the point of closest approach?
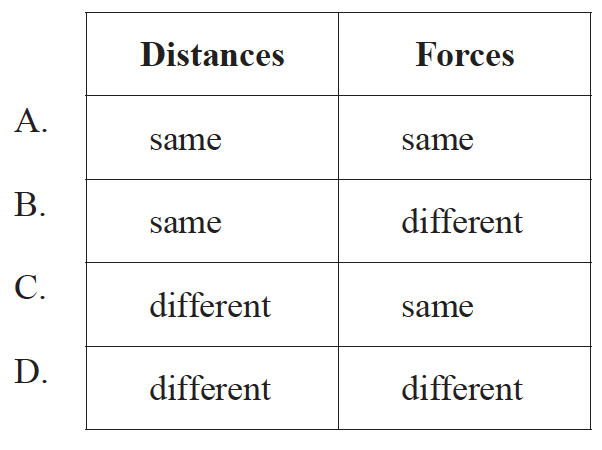
▶️Answer/Explanation
Markscheme
A
Examiners report
There was much discussion of this question on the G2’s. Candidates are expected to choose the best answer and to discount factors that are negligible. The evidence would suggest that many candidates were randomly guessing the answer.
If the difference in the situations is that two isotopes of iron are used, then we can assume the charge is the same and that the distance and force remain unchanged. The slight difference in mass will not affect this.
Question
Which of the following is a correct list of particles upon which the strong nuclear force may act?
A. protons and neutrons
B. protons and electrons
C. neutrons and electrons
D. protons, neutrons and electrons
▶️Answer/Explanation
Markscheme
A
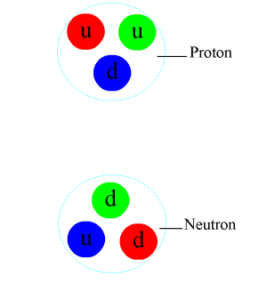
The strong force is held responsible for binding quarks together to form protons, neutrons and other (relatively) heavy particles. This is a short-range force which becomes insignificant at particle separations greater than \(10^-{15 } \)m. The nuclear force between nucleons is thought to be a residual effect of the much stronger force holding quarks together within each nucleon.
