IB DP Physics Mock Exam HL Paper 1B Set 1 - 2025 Syllabus
IB DP Physics Mock Exam HL Paper 1B Set 1
Prepare for the IB DP Physics Mock Exam HL Paper 1B Set 1 with our comprehensive mock exam set 1. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
| Volume | \((10.6 \pm 0.2)\,cm^{3}\) |
| Mass | \((10.82 \pm 0.01)\,g\) |
(ii) Give your final answers in \(kg\,m^{-3}\), using an appropriate number of significant figures.
▶️ Answer/Explanation
(a)
To ensure accuracy, the student should read the volume at the bottom of the meniscus. Alternatively, ensure the cylinder is placed on a horizontal surface so it is vertical, or ensure eyes are level with the reading to avoid parallax error.
(b)
(i) First, calculate the density \(\rho\):
\[\rho = \frac{m}{V} = \frac{10.82}{10.6} \approx 1.02\,g\,cm^{-3}\]
Next, calculate the uncertainty. The fractional uncertainty in mass \(\frac{0.01}{10.82}\) is negligible compared to the volume. We use the fractional uncertainty of the volume:
\[\frac{\Delta \rho}{\rho} \approx \frac{\Delta V}{V} = \frac{0.2}{10.6}\]
\[\Delta \rho = 1.02 \times \frac{0.2}{10.6} \approx 0.019 \approx 0.02\,g\,cm^{-3}\]
So, \(\rho = (1.02 \pm 0.02)\,g\,cm^{-3}\).
(ii) Converting to \(kg\,m^{-3}\) (multiplying by \(1000\)):
\[\rho = 1020 \pm 20\,kg\,m^{-3}\]
The answer is stated to the correct precision (matching the uncertainty).
(c)
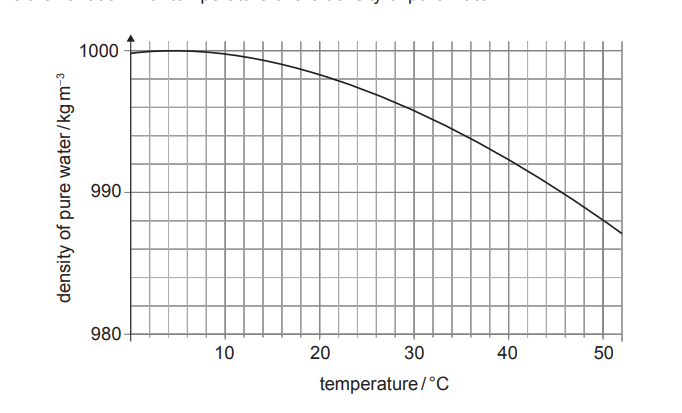
Using the provided graph, the density of pure water at \(35^{\circ}C\) is approximately \(994 \pm 1\,kg\,m^{-3}\). The calculated density of the sample is \(1020 \pm 20\,kg\,m^{-3}\) (range \(1000\) to \(1040\)). Since the value for pure water lies outside the uncertainty range of the sample, the sample is not pure.
