IB DP Physics Mock Exam HL Paper 1B Set 4 - 2025 Syllabus
IB DP Physics Mock Exam HL Paper 1B Set 4
Prepare for the IB DP Physics Mock Exam HL Paper 1B Set 4 with our comprehensive mock exam set 4. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
| \(P / kPa\) | \(V / 10^{-5}m^{3}\) | \(\frac{1}{V} / 10^{4}m^{-3}\) |
|---|---|---|
| \(117\) | \(1.37\) | \(7.30\) |
| \(110\) | \(1.42\) | \(7.04\) |
| \(103\) | \(1.54\) | — |
| \(94\) | \(1.70\) | \(5.88\) |
| \(90\) | \(1.77\) | \(5.65\) |
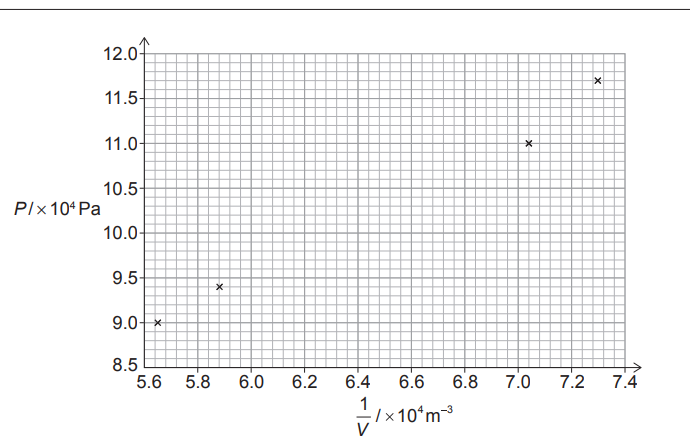
(ii) Draw a suitable line of best fit.
(iii) Describe how the graph can be used to assess whether the relationship \(PV = K\) is supported by the data.
(ii) Give an appropriate SI unit for \(K\).
▶️ Answer/Explanation
(a)
(i) Calculate \(\frac{1}{V}\) for the missing value: \(\frac{1}{1.54 \times 10^{-5}} \approx 6.49 \times 10^{4}\,m^{-3}\).
Plot the point at \((6.5 \times 10^{4}, 10.3 \times 10^{4})\) on the graph.
(ii) Draw a single straight best-fit line so that the data points are evenly distributed about it.
(iii) If \(PV = K\), then \(P = K \times \frac{1}{V}\), so the graph should be a straight line passing through the origin.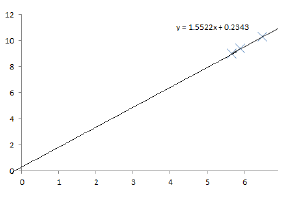
(b)
(i) The value of \(K\) is equal to the gradient of the best-fit line:
\[K = \frac{\Delta P}{\Delta (1/V)} \approx 1.56\]
(ii) Suitable SI units include joules (\(J\)), newton metres (\(Nm\)), or pascal cubic metres (\(Pa\,m^{3}\)).
(c)
Using \(PV = Nk_{B}T\) and \(PV = K\):
\[N = \frac{K}{k_{B}T}\]
Substituting \(K \approx 1.56\), \(T = 291\,K\), and \(k_{B} = 1.38 \times 10^{-23}\,J\,K^{-1}\):
\[N \approx 3.96 \times 10^{20}\]
The number of molecules is approximately \(4.0 \times 10^{20}\).
