IB DP Physics Mock Exam SL Paper 1B Set 2 - 2025 Syllabus
IB DP Physics Mock Exam SL Paper 1B Set 2
Prepare for the IB DP Physics Mock Exam SL Paper 1B Set 2 with our comprehensive mock exam set 2. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
| \(P / kPa\) | \(V / 10^{-5}m^{3}\) | \(\frac{1}{V} / 10^{4}m^{-3}\) |
|---|---|---|
| \(117\) | \(1.37\) | \(7.30\) |
| \(110\) | \(1.42\) | \(7.04\) |
| \(103\) | \(1.54\) | — |
| \(94\) | \(1.70\) | \(5.88\) |
| \(90\) | \(1.77\) | \(5.65\) |
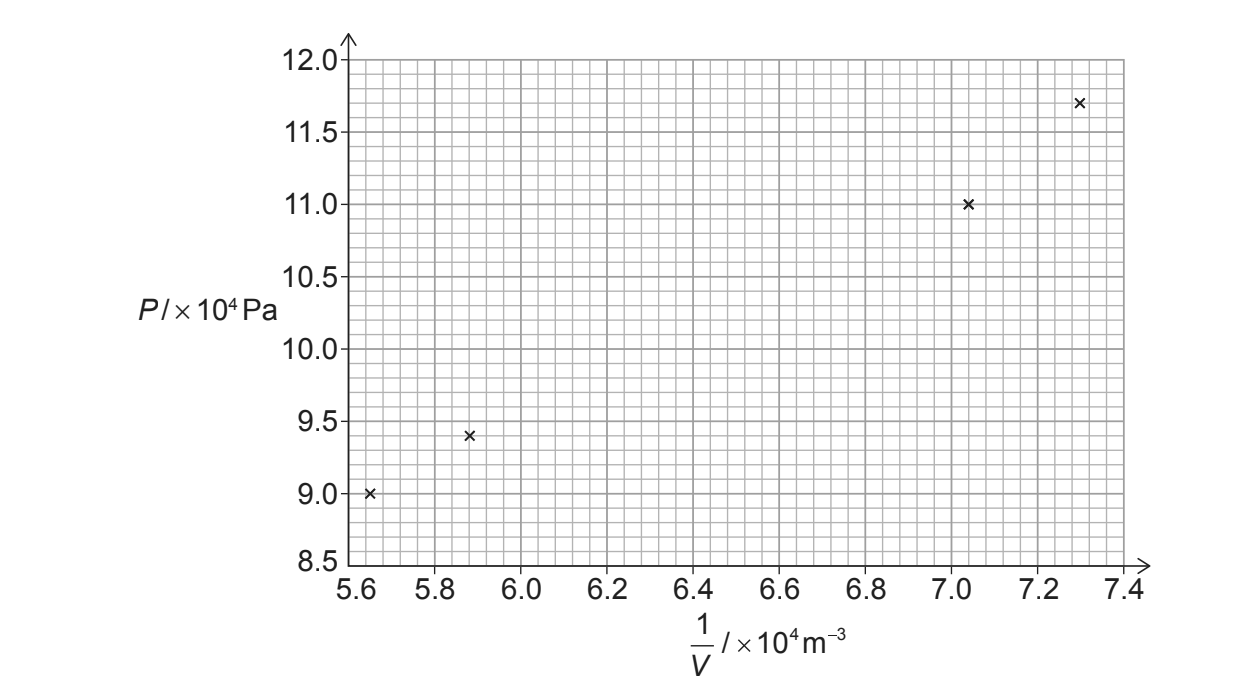
(ii) Draw a suitable line of best fit.
(iii) Describe how the graph can be used to check whether the data are consistent with the relationship \(PV = K\).
(ii) Give an appropriate SI unit for \(K\).
▶️ Answer/Explanation
(a)
(i) Calculate \(\frac{1}{V}\) for the missing point: \(\frac{1}{1.54 \times 10^{-5}} \approx 6.49 \times 10^{4}\,m^{-3}\).
Plot the point at \((6.5 \times 10^{4}, 10.3 \times 10^{4})\) on the graph (within half a square) .
(ii) A clear, single straight line should be drawn using a ruler, ensuring the points are balanced about the line.
(iii) If the relationship \(PV = K\) holds, then \(P = K \times \frac{1}{V}\). Therefore, the graph of \(P\) versus \(\frac{1}{V}\) should be a straight line that passes through (or is close to) the origin. Alternatively, one could evaluate \(PV\) for each data point to confirm they are approximately constant.
(b)
(i) Determine the gradient of the best-fit line using two points on the line. Alternatively, calculate the average \(PV\) for the data points.
\(K = \text{gradient} \approx 1.56\) (Accept range \(1.55 – 1.60\)) .
(ii) The unit is Joules (\(J\)), \(Nm\), or \(Pa\,m^{3}\) .
(c)
Using the ideal gas equation \(PV = Nk_{B}T\), we know that \(PV = K\), so \(K = Nk_{B}T\) .
Rearranging for \(N\) (number of molecules):
\(N = \frac{K}{k_{B}T}\)
Using \(K \approx 1.59\) (or the value calculated in b(i)), \(T = 291\,K\), and Boltzmann’s constant \(k_{B} \approx 1.38 \times 10^{-23}\,J\,K^{-1}\):
\(N = \frac{1.59}{1.38 \times 10^{-23} \times 291} \approx 3.96 \times 10^{20}\) .
Answer: \(3.96 \times 10^{20}\) molecules (approx \(4.0 \times 10^{20}\)).
