A positive pion decays into a positive muon and a neutrino.
\[{\pi ^ + } \to {\mu ^ + } + {v_\mu }\]
The momentum of the muon is measured to be 29.8 MeV c–1 in a laboratory reference frame in which the pion is at rest. The rest mass of the muon is 105.7 MeV c–2 and the mass of the neutrino can be assumed to be zero.
For the laboratory reference frame
a.i.write down the momentum of the neutrino.[1]
▶️Answer/Explanation
Markscheme
a.i.
«–»29.8 «MeVc–1»
[1 mark]
Eπ = \(\sqrt {p_\mu ^2{c^2} + m_\mu ^2{c^4}} \) + pvc OR Eμ = 109.8 «MeV»
Eπ = «\(\sqrt {{{29.8}^2} + {{105.7}^2}} \) + 29.8 =» 139.6 «MeV»
Final value to at least 3 sig figs required for mark.
[2 marks]
139.6 MeVc–2
Units required.
Accept 140 MeVc–2.
[1 mark]
A student pours a canned carbonated drink into a cylindrical container after shaking the can violently before opening. A large volume of foam is produced that fills the container. The graph shows the variation of foam height with time.
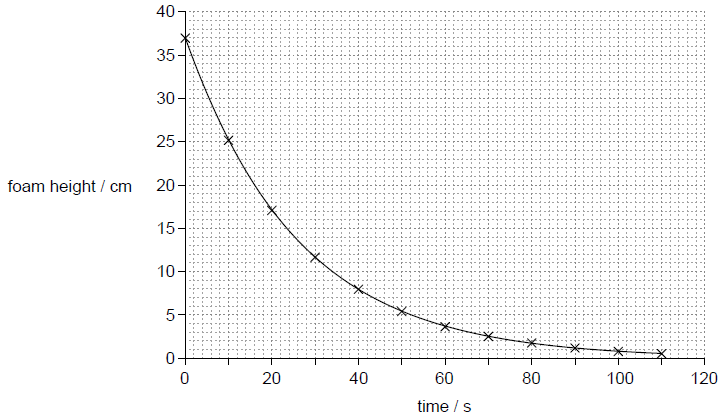
a.Determine the time taken for the foam to drop to
(i) half its initial height.
(ii) a quarter of its initial height.[2]
▶️Answer/Explanation
Markscheme
a.
i
18 «s»
Allow answer in the range of 17 «s» to 19 «s».
Ignore wrong unit.
ii
36 «s»
Allow answer in the range of 35 «s» to 37 «s».
radioactive/nuclear decay
OR
capacitor discharge
OR
cooling
Accept any relevant situation, eg: critically damping, approaching terminal velocity
