IB PHYSICS HL(Higher level) – 2024 – Practice Questions- All Topics
Topic 3.2 Modelling a gas
Topic 1 Weightage : 3 %
All Questions for Topic 3.2 –Pressure , Equation of state for an ideal gas , Kinetic model of an ideal gas , Mole, molar mass and the Avogadro constant , Differences between real and ideal gases
Question
The air in a kitchen has pressure 1.0 × 105 Pa and temperature 22°C. A refrigerator of internal volume 0.36 m3 is installed in the kitchen.
With the door open the air in the refrigerator is initially at the same temperature and pressure as the air in the kitchen. Calculate the number of molecules of air in the refrigerator. [2]
The refrigerator door is closed. The air in the refrigerator is cooled to 5.0°C and the number of air molecules in the refrigerator stays the same.
(i) Determine the pressure of the air inside the refrigerator. [2]
(ii) The door of the refrigerator has an area of 0.72 m2. Show that the minimum force needed to open the refrigerator door is about 4 kN. [2]
(iii) Comment on the magnitude of the force in (b)(ii). [2]
▶️Answer/Explanation
Ans:
a N = \(\frac{pv}{kT}\) OR N = \(\frac{1.0\times 10\times ^5\times 0.36}{1.38\times 10^{-23\times 295}}\) N = 8.8 ×10 24
b i use of \(\frac{P}{T}\) = constant OR P = \(\frac{nRT}{V}\) OR \(\frac{NkT}{V}\) p = 9.4× 104 «Pa »
b ii F = A p = ×∆ F = 0.72 × (1.0 – 0.94) ×105 OR 4.3 ×103 «N »
b iii force is «very» large there must be a mechanism that makes this force smaller OR assumption used to calculate the force/pressure is unrealistic
Question
A mass of 1.0 kg of water is brought to its boiling point of 100 °C using an electric heater of power 1.6 kW.
(a) (i) The molar mass of water is 18 g mo l-1. Estimate the average speed of the water molecules in the vapor produced. Assume the vapor behaves as an ideal gas. [2]
(ii) State one assumption of the kinetic model of an ideal gas. [1]
(b) A mass of 0.86 kg of water remains after it has boiled for 200 s.
(i) Estimate the specific latent heat of vaporization of water. State an appropriate unit for your answer. [2]
(ii) Explain why the temperature of water remains at 100 °C during this time. [1]
(c) The heater is removed and a mass of 0.30 kg of pasta at –10 °C is added to the boiling water.
Determine the equilibrium temperature of the pasta and water after the pasta is added. Other heat transfers are negligible.
Specific heat capacity of pasta = 1.8 kJ k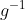
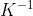
Specific heat capacity of water = 4.2 kJ k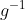
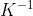 [3]
[3]
(d) The electric heater has two identical resistors connected in parallel.
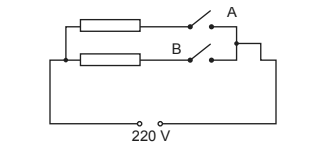
The circuit transfers 1.6 kW when switch A only is closed. The external voltage is 220 V.
(i) Show that each resistor has a resistance of about 30 Ω. [1]
(ii) Calculate the power transferred by the heater when both switches are closed. [2]
▶️Answer/Explanation
Ans:
a i Ek= «\(\frac{3}{2}(1.38 \times 10a^{-23)}(373)\)» = 7.7 ×10-21 « J» V = « \(\sqrt{\frac{3\times 1.38 \times 10 a^{-23 }\times 373}{0.018}}\)
ii particles can be considered points «without dimensions» no intermolecular forces/no forces between particles «except during collisions» the volume of a particle is negligible compared to volume of gas collisions between particles are elastic time between particle collisions are greater than time of collision no intermolecular PE/no PE between particles
b i «mL = P t» so «L = \(\frac{1600\times 200}{0.14}\) » = 2.3 x 106 «J kg-1» J kg-1
b ii «all» of the energy added is used to increase the «intermolecular» potential energy of the particles/break «intermolecular» bonds/OWTTE Accept reference to atoms/molecules for “particle”
c use of mc∆T 0.86×4200× (100 – T) = 0.3×1800× (T +10) Teq = 85.69«°C» ≅ 86«°C»
d i P = \(\frac{Va^{2}}{R}\) so \(\frac{220a^{2}}{1600}\) so R = 30.25 «Ω »
d ii use of parallel resistors addition so Req = 15 «Ω » P = 3200 «W»
