IB MYP Chemistry Mock Test 2 – 2026 Edition
IB MYP Chemistry Mock Test 2 – April/May 2026 Exam
IB MYP Chemistry Mock Test 2: Prepare for the MYP exams with subject-specific Prediction questions, model answers. All topics covered.
Prepared by MYP teachers: Access our IB MYP Chemistry Mock Test 2 Mock with model answer. Students: Practice with exam-style papers for MYP Exam
Question : Hard Water Chemistry
Natural sources of water can contain different dissolved ions. Water containing mainly group 2 ions such as calcium and magnesium (Ca²⁺ and Mg²⁺) is known as “hard” water.

Question a [2 marks] – Matter: Atomic Structure
State the number of protons and neutrons in an ²⁴Mg²⁺ ion.
Protons:
Neutrons:
▶️ Answer/Explanation
Protons: 12
Neutrons: 12
Explanation: The atomic number of Mg is 12 (protons). Mass number is 24, so neutrons = 24 – 12 = 12. The 2+ charge doesn’t affect proton/neutron count.
Question b [1 mark] – Bonding: Chemical Formulas
Determine the formula of magnesium phosphate.
▶️ Answer/Explanation
Mg₃(PO₄)₂
Explanation: Phosphate ion is PO₄³⁻ and magnesium ion is Mg²⁺. To balance charges, we need 3 Mg²⁺ ions (total +6) and 2 PO₄³⁻ ions (total -6).
Question c [4 marks] – Periodic Table: Metals and Non-metals
Identify two physical properties that distinguish between magnesium and chlorine at 25°C.
Property 1:
How this property distinguishes between magnesium and chlorine:
Property 2:
How this property distinguishes between magnesium and chlorine:
▶️ Answer/Explanation
Property 1: State/Phase
Magnesium is a solid at 25°C while chlorine is a gas.
Property 2: Electrical Conductivity
Magnesium (metal) conducts electricity while chlorine (non-metal) does not.
Alternative properties could include:
- Luster: Mg is shiny, Cl₂ is not
- Malleability: Mg is malleable, Cl₂ is not
- Melting/Boiling points: Mg has much higher values
Question d [1 mark] – Bonding: Structure and Bonding
State the type of bond that would form in a reaction between magnesium and chlorine.
▶️ Answer/Explanation
Ionic bond
Explanation: Magnesium (metal) donates electrons to chlorine (non-metal), forming Mg²⁺ and Cl⁻ ions that attract each other through ionic bonding.
Question e [4 marks] – Pure and Impure Substance: Separation Techniques
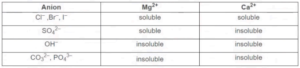
Using the solubility data, explain how you would remove Ca²⁺ ions only from hard water.
▶️ Answer/Explanation
- Add sulfate ions (e.g., Na₂SO₄) to the water
- Calcium sulfate is insoluble and will precipitate out
- Magnesium sulfate remains soluble in solution
- Filter the mixture to remove the calcium sulfate precipitate
Explanation: The solubility table shows CaSO₄ is insoluble while MgSO₄ is soluble, allowing selective removal of Ca²⁺ ions.
Question f [1 mark] – Bonding: The Mole Concept
Crystals of magnesium sulphate pentahydrate (MgSO₄·5H₂O) are heated to form magnesium sulphate. State the number of moles of water formed if 0.5 moles of MgSO₄·5H₂O are heated.
▶️ Answer/Explanation
2.5 moles
Explanation: Each mole of MgSO₄·5H₂O releases 5 moles of water. For 0.5 moles: 0.5 × 5 = 2.5 moles of water.
