IB MYP Physics Mock Tests 1 – 2026 Edition
IB MYP Physics Mock Tests 1 – April/May 2026 Exam
IB MYP Physics Mock Tests 1: Prepare for the MYP exams with subject-specific Prediction questions, model answers. All topics covered.
Prepared by MYP teachers: Access our IB MYP Physics Mock Tests 1 Mock with model answer. Students: Practice with exam-style papers for MYP Exam
Question : Coal-Fired Power Stations [12 marks]
a Question a [1 mark] – States of Matter
A coal-fired power station involves three states of matter. Identify the location of each of the substances below:
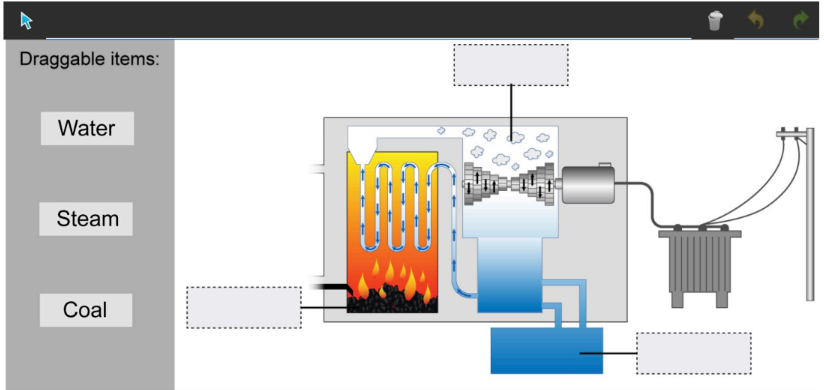
Draggable items:
- Water
- Steam
- Coal
▶️Answer/Explanation
Solution:
1. Water is in the liquid state, typically found in the boiler where it is heated.
2. Steam is the gaseous state of water, formed when water is heated to its boiling point in the boiler.
3. Coal is in the solid state, burned in the combustion chamber to release chemical potential energy.
b Question b [1 mark] – Kinetic Energy
Absorbing heat can affect the kinetic energy of water molecules. State how water molecules are affected by this increase in kinetic energy.
▶️Answer/Explanation
Solution:
When water absorbs heat, the kinetic energy of its molecules increases. This causes the molecules to move faster, leading to an increase in temperature. If enough heat is absorbed, the water may eventually change state from liquid to gas (steam).
c Question c [2 marks] – Heat Energy and Phase Change
As water boils into steam, the heat energy absorbed does not affect the kinetic energy of the molecules. Outline what happens to this heat energy referring to the movement and arrangement of particles.
▶️Answer/Explanation
Solution:
During boiling, the heat energy absorbed is used to overcome the intermolecular forces between water molecules rather than increasing their kinetic energy. This energy breaks the hydrogen bonds, changing the arrangement of particles from a closely packed liquid to a more dispersed gas (steam). The movement of particles becomes more random and less restricted.
d Question d [2 marks] – Energy Calculation
One kilogram of coal contains approximately \( 2.5 \times 10^7 \, \text{J} \) of chemical potential energy. For a coal-fired power station that burns \( 4.5 \times 10^5 \, \text{kg} \) of coal per hour, calculate the energy released in one hour. You should give your answer to two significant figures.
▶️Answer/Explanation
Solution:
Energy released = Energy per kg × Mass of coal burned
\[ \text{Energy} = 2.5 \times 10^7 \, \text{J/kg} \times 4.5 \times 10^5 \, \text{kg} \]
\[ \text{Energy} = 1.125 \times 10^{13} \, \text{J} \]
Rounded to two significant figures: \( 1.1 \times 10^{13} \, \text{J} \).
e Question e [2 marks] – Power Output Calculation
Another coal-fired power station burns enough coal per hour to generate a power of 26 700 MW. If the efficiency of this power station is 28%, determine the useful power output.
▶️Answer/Explanation
Solution:
Useful power output = Total power × Efficiency
\[ \text{Useful power} = 26\,700 \, \text{MW} \times 0.28 \]
\[ \text{Useful power} = 7\,476 \, \text{MW} \]
Thus, the useful power output is 7,476 MW.
f Question f [1 mark] – Greenhouse Gases
Burning coal contributes to climate change by altering the composition of Earth’s atmosphere, increasing the quantity of greenhouse gases. The diagram below shows the flow of energy through the Earth’s atmosphere.
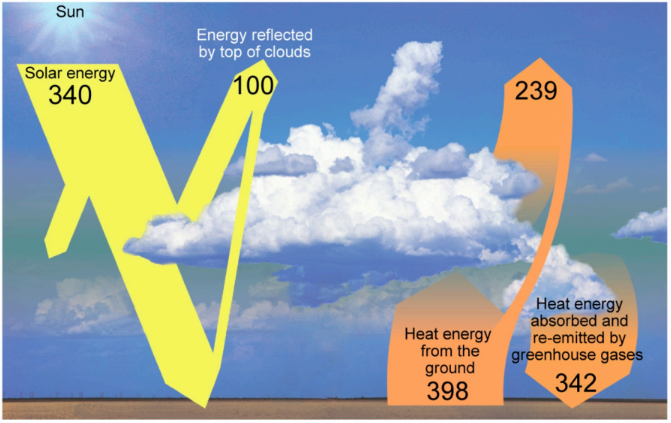
Select the arrow on the diagram which would be most affected by burning coal.
- Solar energy
- Energy reflected by top of clouds
- Heat energy from the ground
- Heat energy absorbed and re-emitted by greenhouse gases
▶️Answer/Explanation
Solution:
The correct answer is “Heat energy absorbed and re-emitted by greenhouse gases”.
Burning coal releases CO₂ and other greenhouse gases, which trap infrared radiation (heat) emitted by the Earth’s surface. This increases the amount of heat energy absorbed and re-emitted by greenhouse gases, enhancing the greenhouse effect.
g Question g [3 marks] – Climate Change Impact
Greenhouse gases trap heat energy in Earth’s atmosphere. This trapped heat is associated with more extreme weather events like storms, droughts, floods and changes to Earth’s climate. Using scientific knowledge, explain why trapping heat energy in Earth’s atmosphere would cause more extreme weather events and changes to Earth’s climate.
▶️Answer/Explanation
Solution:
1. Increased Global Temperatures: Trapped heat raises Earth’s average temperature, leading to melting ice caps, rising sea levels, and disrupted ecosystems.
2. Energy Imbalance: More heat energy in the atmosphere fuels stronger convection currents, intensifying storms, hurricanes, and rainfall patterns.
3. Disrupted Weather Systems: Warmer oceans evaporate more water, increasing humidity and precipitation in some regions while causing droughts in others due to shifting air currents (e.g., jet streams).
4. Feedback Loops: Example: Melting permafrost releases methane (a potent greenhouse gas), further accelerating warming.
Syllabus Reference
Unit 1: Energy
- States of matter and phase changes
- Energy transformations and efficiency
- Power calculations
Unit 2: Climate Science
- Greenhouse effect
- Impact of human activities on climate
Assessment Criteria: A (Knowledge and understanding), B (Inquiring and designing), & C (Processing and evaluating)
