IB MYP Physics Mock Tests 4 – 2026 Edition
IB MYP Physics Mock Tests 4 – April/May 2026 Exam
IB MYP Physics Mock Tests 4: Prepare for the MYP exams with subject-specific Prediction questions, model answers. All topics covered.
Prepared by MYP teachers: Access our IB MYP Physics Mock Tests 4 Mock with model answer. Students: Practice with exam-style papers for MYP Exam
Question : States and properties of matter, kinetic theory [7 marks]
1905 is known as Einstein’s annus mirabilis (wonderful year) as he made several discoveries that changed our understanding of the physical world.
One of the consequences of this work is kinetic theory. The theory explains the physical properties of matter. The theory can be used to explain why matter exists in different states (solid, liquid, and gas) and how matter can change from one state to the next.
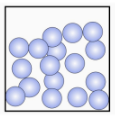
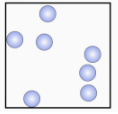
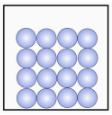
a Select the states of matter represented by each image. [1 mark]
▶️Answer/Explanation
Correct answers:
- Solid (particles in fixed positions with regular arrangement)
- Liquid (particles close together but able to move past each other)
- Gas (particles far apart moving freely)
Key Concept:
The kinetic theory explains states of matter based on particle arrangement and motion. Solids have fixed positions, liquids have mobile particles but fixed volume, and gases have particles that fill their container.
b Select the states of matter to complete the table below. [1 mark]
| Change of state | Initial State | Final State |
|---|---|---|
| Condensation | ||
| Vaporization |
▶️Answer/Explanation
Correct answers:
| Change of state | Initial State | Final State |
|---|---|---|
| Condensation | Gas | Liquid |
| Vaporization | Liquid | Gas |
Key Concept:
Phase changes involve energy transfer. Condensation is gas→liquid (heat released), vaporization is liquid→gas (heat absorbed). These changes occur when particles gain/lose enough kinetic energy to overcome intermolecular forces.
c Use kinetic theory to explain why the balloon expands when the Bunsen burner is lit. [3 marks]
A student conducts an experiment in which she places a balloon over a glass bottle filled with a gas at room temperature. A Bunsen burner is lit and the balloon expands as the temperature of the gas increases.
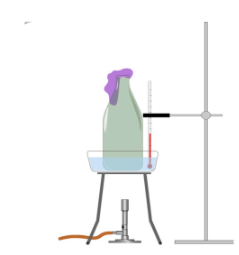
Explanation space:
▶️Answer/Explanation
Key points for full marks:
- Particles gain kinetic energy when heated (move faster)
- Increased particle collisions per second with balloon walls
- Collisions occur with greater force
- Resulting in greater pressure inside the balloon
- Balloon expands to equalize internal and external pressures
Sample Answer:
“When heated, gas particles move faster with greater kinetic energy. They collide more frequently and with more force against the balloon walls, increasing internal pressure. The balloon expands as the increased pressure pushes outward until internal and external pressures balance.”
d Use your knowledge of kinetic theory to suggest the cause of the observed random movement of smoke particles in air. [2 marks]
“Brownian motion is the random movement of small particles suspended in a gas or a liquid. Brownian motion can be seen in smoke particles in air and pollen grains in water.”

Explanation space:
▶️Answer/Explanation
Key points for full marks:
- Air particles are in constant random motion (but too small to be seen)
- These moving air particles collide with larger smoke particles
- The collisions cause the visible random motion of smoke particles
- The motion provides evidence for kinetic theory of gases
Sample Answer:
“Invisible air molecules are constantly moving and colliding with the larger smoke particles from different directions. These random collisions cause the visible jittery motion of smoke particles, demonstrating the kinetic theory of gases in action.”
Syllabus Reference
States and properties of matter
- Particle arrangement in solids, liquids, gases
- Phase changes and energy transfer
Kinetic theory
- Pressure-temperature relationship
- Brownian motion evidence
Thermal physics
- Heat transfer mechanisms
- Thermal expansion
Assessment Criteria: A (Knowledge), C (Communication), D (Scientific Thinking)
