IB MYP Integrated Science- Chemistry- Atomic structure-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Atomic structure -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Atomic structure -Study Notes -As per latest Syllabus.
Key Concepts:
Atomic Structure
IB MYP Integrated Science -Concise Summary Notes- All Topics
Atomic Structure
All matter is made up of atoms, which are the smallest particles of an element that retain its chemical properties. Atoms are composed of smaller particles called subatomic particles, which determine the structure, mass, and behavior of the atom.
Subatomic Particles
There are three main subatomic particles:
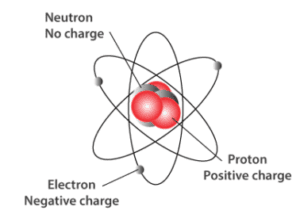
- Protons — positive charge \( (+1) \), located in the nucleus
- Neutrons — no charge \( (0) \), located in the nucleus
- Electrons — negative charge \( (-1) \), found in energy levels around the nucleus
Relative Mass and Charge
- Proton: mass = 1, charge = +1
- Neutron: mass = 1, charge = 0
- Electron: mass = \( \frac{1}{1836} \), charge = -1
Almost all the mass of an atom is concentrated in the nucleus because electrons have a very small mass.
Atomic Number and Mass Number
The atomic number represents the number of protons in an atom: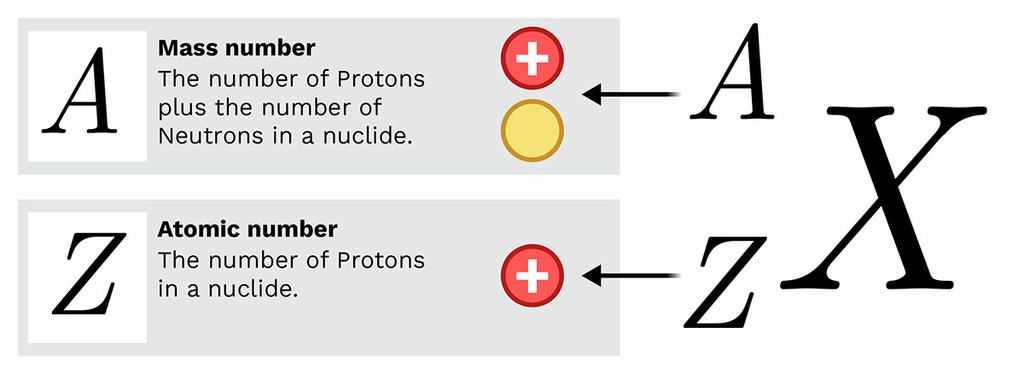
\( \text{Atomic number} = \text{number of protons} \)
In a neutral atom:
\( \text{protons} = \text{electrons} \)
The mass number is the total number of protons and neutrons:
\( \text{Mass number} = \text{protons} + \text{neutrons} \)
So the number of neutrons can be calculated using:
\( \text{neutrons} = \text{mass number} – \text{atomic number} \)
Nuclear Notation (Atomic Representation)
Atoms can be represented using nuclear notation:
\( \mathrm{^{A}_{Z}\text{X}} \)
Where:
\(\rm A \) = mass number (protons + neutrons)
\( \rm Z \) = atomic number (protons)
Example:
\( ^{23}_{11}\text{Na} \)
Electron Arrangement (Bohr Model)
Electrons occupy fixed energy levels (shells) around the nucleus. This model is known as the Bohr model.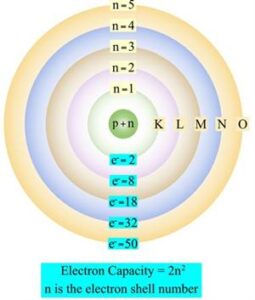
For MYP level:
- First shell: up to 2 electrons
- Second shell: up to 8 electrons
- Third shell: up to 8 electrons
Electrons fill the lowest energy level first before moving to higher levels.
Examples:
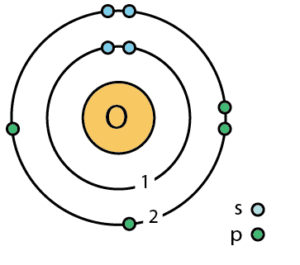
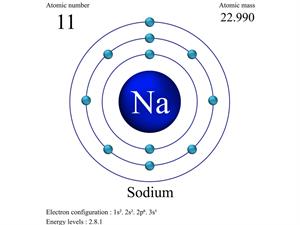
- Oxygen (8): \( 2,6 \)
- Sodium (11): \( 2,8,1 \)
Valence Electrons and Reactivity
The electrons in the outermost shell are called valence electrons. These determine how atoms react and form chemical bonds.
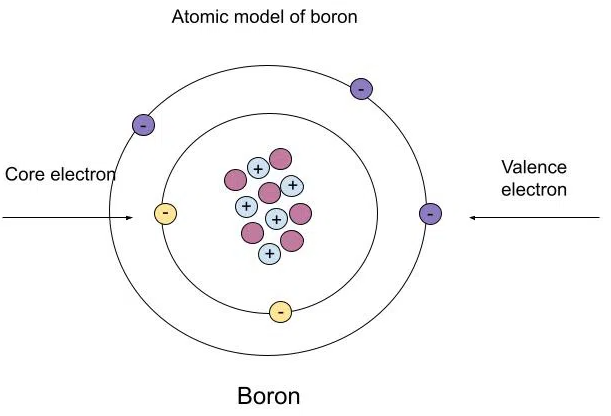
Elements with the same number of valence electrons have similar chemical properties.
Link to the Periodic Table
Elements in the periodic table are arranged in order of increasing atomic number.
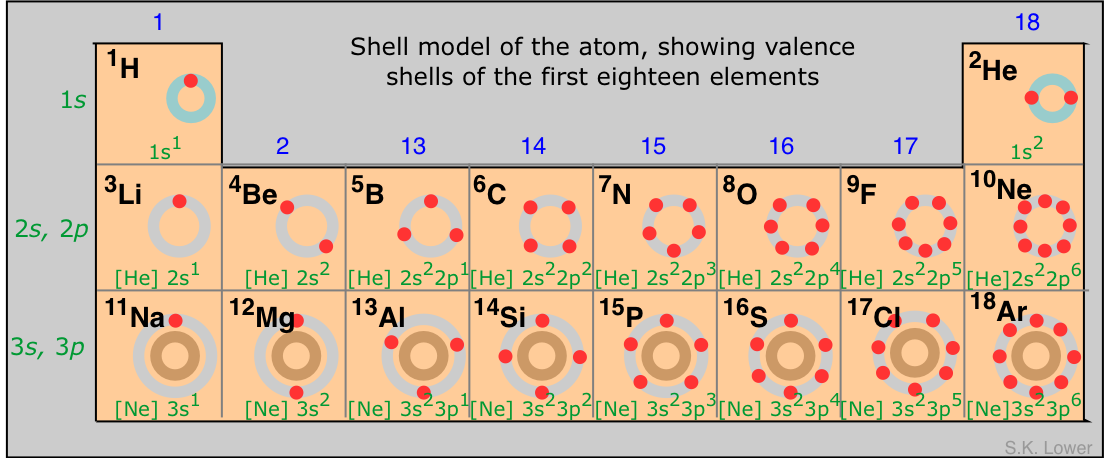
The position of an element tells us:
- Number of shells → period
- Number of valence electrons → group (for main group elements)
Isotopes
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons.
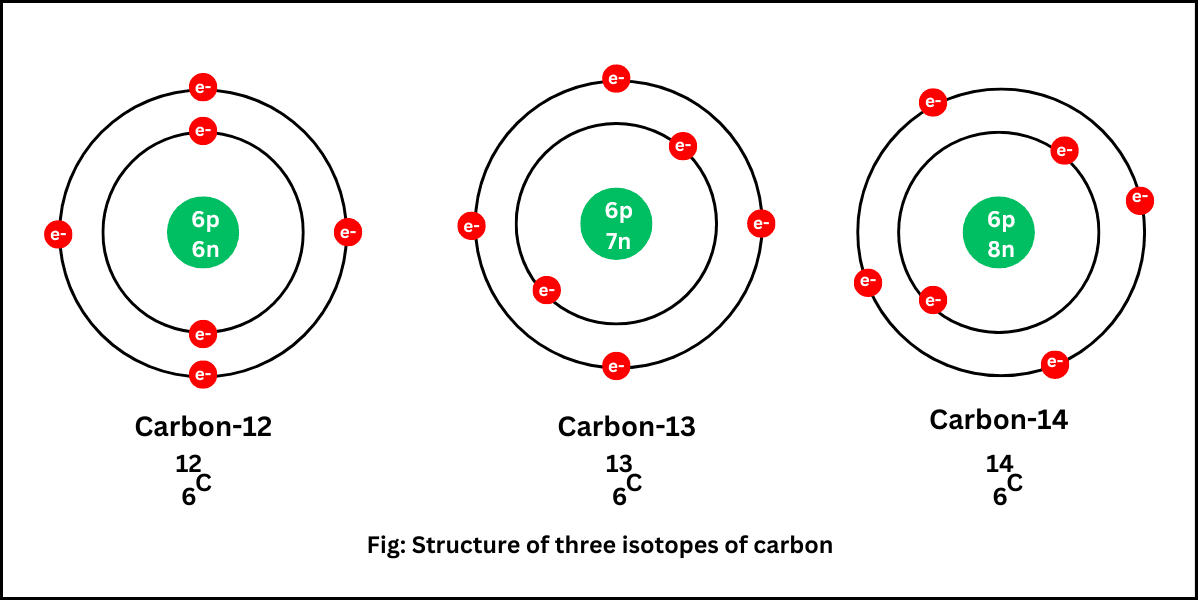
Same atomic number, different mass number
Example:
Carbon-12 and Carbon-14
Applications of Isotopes (IB Context)
- Carbon-14 is used in radiocarbon dating to determine the age of fossils
- Some isotopes are used in medicine for diagnosing and treating diseases
Ions
Atoms can gain or lose electrons to form charged particles called ions.
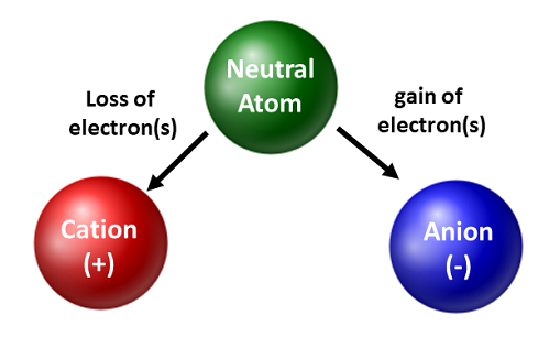
Comparison:
- Neutral atom: protons = electrons
- Ion: protons ≠ electrons
- Loss of electrons → positive ion (cation)
- Gain of electrons → negative ion (anion)
Charge is calculated using:
\( \text{charge} = \text{protons} – \text{electrons} \)
Real-World Importance of Ions
Ions such as \( \text{Na}^+ \) and \( \text{K}^+ \) are essential for nerve signal transmission in the human body
Example 1:
An atom is represented as \( ^{31}_{15}\text{P} \). Determine the number of protons, neutrons, electrons, and write its electron arrangement.
▶️ Answer/Explanation
Protons = 15
Electrons = 15
Neutrons = \( 31 – 15 = 16 \)
Electron arrangement = \( 2,8,5 \)
Conclusion: The atom has 15 protons, 16 neutrons, and configuration \( 2,8,5 \).
Example 2:
An ion is written as \( ^{24}_{12}\text{Mg}^{2+} \). Determine the number of protons, neutrons, and electrons.
▶️ Answer/Explanation
Protons = 12
Neutrons = \( 24 – 12 = 12 \)
Electrons = \( 12 – 2 = 10 \)
Conclusion: The ion has 12 protons, 12 neutrons, and 10 electrons.
Example 3 :
An ion is represented as \( \mathrm{^{56}_{26}Fe^{3+}} \).
a) Determine the number of protons, neutrons, and electrons.
b) Write the electron arrangement of the neutral atom and the ion.
c) Explain how the ion is formed.
▶️ Answer/Explanation
Step 1: Protons
Protons = 26
Step 2: Neutrons
\( 56 – 26 = 30 \)
Step 3: Electrons (Ion)
\( 26 – 3 = 23 \)
Step 4: Electron Arrangement
Neutral atom (Fe):
\( 2,8,14,2 \)
Ion \( \mathrm{Fe^{3+}} \):
\( 2,8,13 \)
Step 5: Formation of Ion
The iron atom loses 3 electrons from its outer energy levels to achieve a more stable configuration.
Conclusion: The ion has 26 protons, 30 neutrons, 23 electrons, and is formed by losing 3 electrons.
