IB MYP Integrated Science- Chemistry- Chemical bonding-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Chemical bonding -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Chemical bonding -Study Notes -As per latest Syllabus.
Key Concepts:
Chemical bonding (ionic, covalent, and metallic bonding)
IB MYP Integrated Science -Concise Summary Notes- All Topics
Chemical Bonding (Ionic, Covalent, and Metallic Bonding)
Chemical bonding is the process by which atoms combine to form compounds. Atoms bond together to achieve a more stable electron configuration, usually by obtaining a full outer shell of electrons.
Why Atoms Bond
Atoms are more stable when their outer shell is full. This is often referred to as achieving a noble gas configuration.
Atoms can achieve this by:
- Losing electrons
- Gaining electrons
- Sharing electrons
Ionic Bonding
Ionic bonding occurs when electrons are transferred from one atom to another, forming oppositely charged ions that attract each other.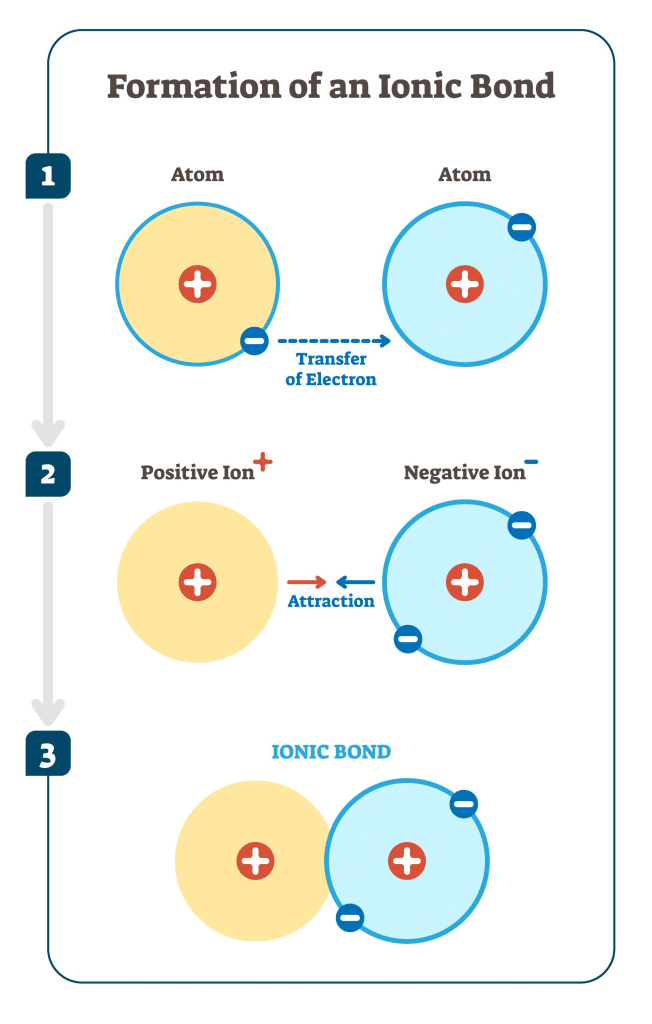
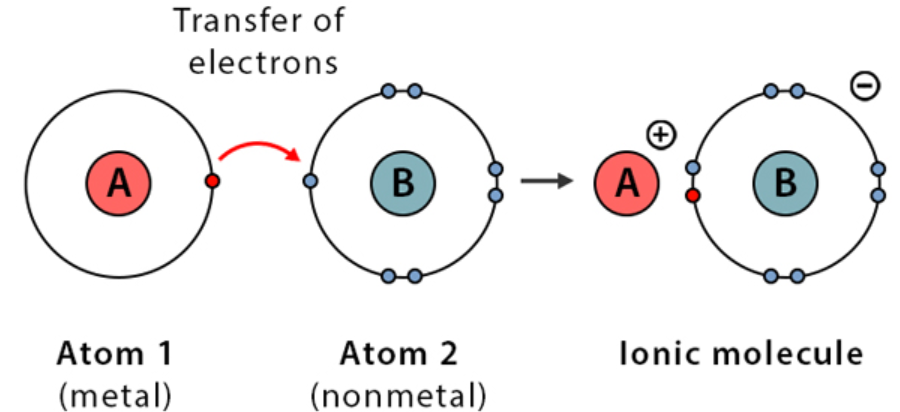
This usually happens between a metal and a non-metal.
Example:
\( \mathrm{Na \rightarrow Na^+ + e^-} \)
\( \mathrm{Cl + e^- \rightarrow Cl^-} \)
The oppositely charged ions attract to form an ionic compound:
\( \mathrm{Na^+ + Cl^- \rightarrow NaCl} \)
Properties of Ionic Compounds
- High melting and boiling points
- Conduct electricity when molten or dissolved in water
- Form crystal lattice structures
Covalent Bonding
Covalent bonding occurs when atoms share pairs of electrons to achieve a full outer shell.
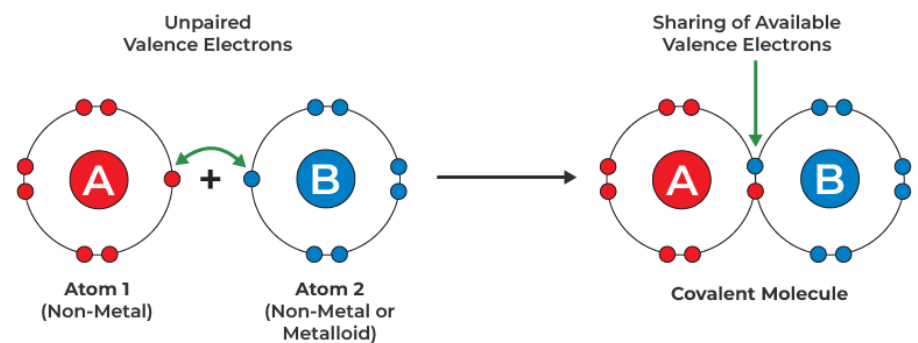
This usually occurs between non-metals.
Examples:
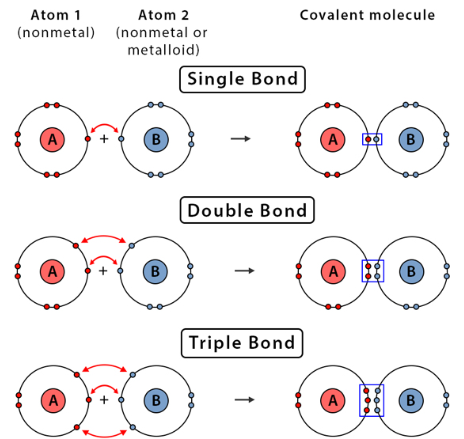
- \( \mathrm{H_2} \) (single bond)
- \( \mathrm{O_2} \) (double bond)
- \( \mathrm{N_2} \) (triple bond)
Properties of Covalent Substances
- Usually low melting and boiling points (simple molecules)
- Do not conduct electricity
- Can form gases, liquids, or solids
Metallic Bonding
Metallic bonding occurs in metals where atoms release electrons to form a sea of delocalised electrons surrounding positive metal ions.
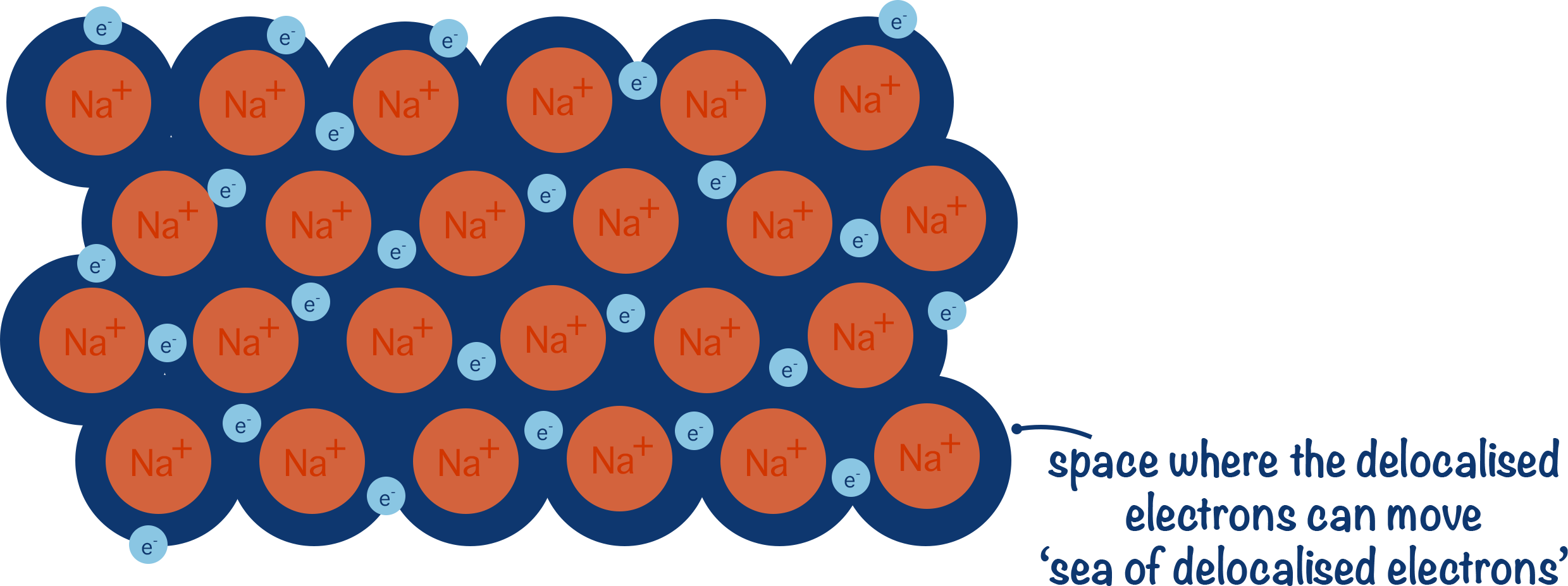
These free electrons can move throughout the structure.
Properties of Metals
- Good conductors of electricity and heat
- Malleable (can be shaped)
- Ductile (can be drawn into wires)
Comparison of Bonding Types
- Ionic: transfer of electrons (metal + non-metal)
- Covalent: sharing of electrons (non-metals)
- Metallic: delocalised electrons in a metal lattice
Real-World Applications
- Ionic compounds like \( \mathrm{NaCl} \) are used in food and industry
- Covalent substances like water are essential for life
- Metals are used in electrical wiring due to conductivity
Example 1:
Explain how sodium and chlorine form an ionic bond.
▶️ Answer/Explanation
Sodium loses 1 electron: \( \mathrm{Na \rightarrow Na^+ + e^-} \)
Chlorine gains 1 electron: \( \mathrm{Cl + e^- \rightarrow Cl^-} \)
Opposite charges attract to form \( \mathrm{NaCl} \)
Conclusion: Ionic bonding occurs through electron transfer and electrostatic attraction.
Example 2:
Describe the bonding in \( \mathrm{H_2O} \).
▶️ Answer/Explanation
Oxygen shares electrons with two hydrogen atoms
Each bond is a shared pair of electrons
Conclusion: Water has covalent bonding with shared electron pairs.
Example 3 :
A substance has a high melting point, conducts electricity when molten, and is brittle. Identify the type of bonding and explain your reasoning.
▶️ Answer/Explanation
Step 1: Identify properties
High melting point → strong bonds
Conducts when molten → mobile ions
Brittle → lattice structure
Step 2: Conclusion
This is an ionic compound
Explanation: Ionic compounds have strong electrostatic forces, allowing conduction when ions are free to move.
