IB MYP Integrated Science- Chemistry- Combustion-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Combustion -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Combustion -Study Notes -As per latest Syllabus.
Key Concepts:
Combustion
IB MYP Integrated Science -Concise Summary Notes- All Topics
Combustion
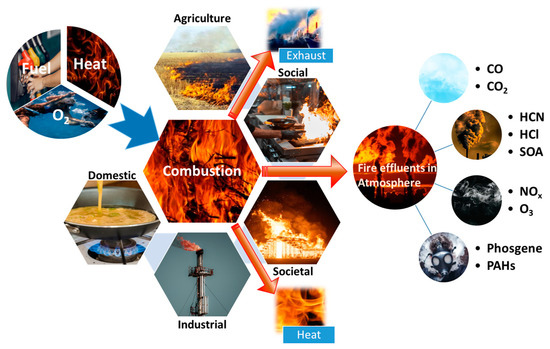
Combustion is a chemical reaction in which a substance (fuel) reacts with oxygen to release energy in the form of heat and often light. It is an exothermic reaction because energy is released to the surroundings.
General Equation of Combustion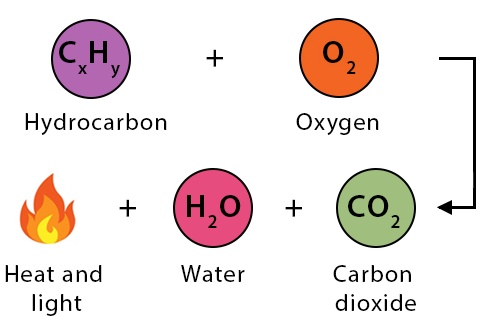
\( \mathrm{Fuel + O_2 \rightarrow CO_2 + H_2O + energy} \)
For example, combustion of methane:
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Conditions Required for Combustion
Combustion requires three essential components, known as the fire triangle:
- Fuel (substance that burns)
- Oxygen (supports burning)
- Heat (ignition temperature)
Removing any one of these will stop the fire.
Types of Combustion
Complete Combustion
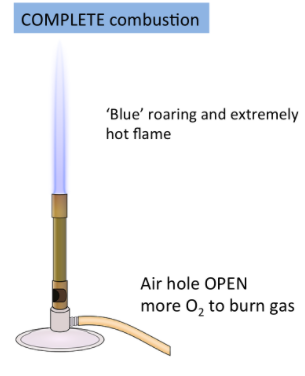
- Occurs with sufficient oxygen
- Produces carbon dioxide and water
- Clean blue flame
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Incomplete Combustion
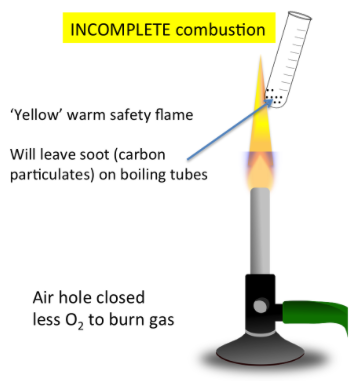
- Occurs with limited oxygen
- Produces carbon monoxide (CO) or carbon (soot)
- Yellow/orange smoky flame
\( \mathrm{CH_4 + O_2 \rightarrow CO + H_2O} \)
Other Types of Combustion
- Rapid combustion: burns quickly with flame
- Slow combustion: occurs slowly without flame (e.g., respiration)
- Spontaneous combustion: occurs without external ignition (e.g., oily rags)
Energy Changes in Combustion
Combustion reactions release large amounts of energy due to bond breaking and formation.
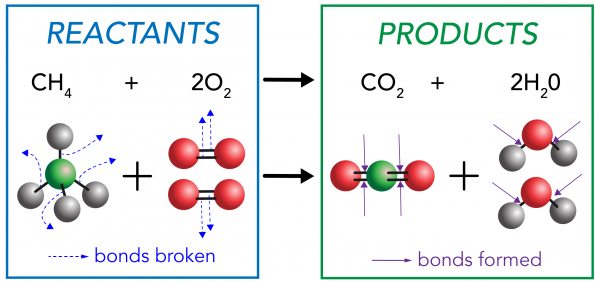
\( \mathrm{Energy\ released > energy\ absorbed} \)
Environmental Impact
- Carbon dioxide contributes to global warming

- Carbon monoxide is toxic
- Soot causes air pollution and respiratory problems
Improving Combustion Efficiency
- Ensure good oxygen supply
- Use finely divided fuel (larger surface area)
- Maintain proper temperature
Real-World Applications
- Engines use combustion to produce energy
- Cooking and heating systems rely on combustion
- Power plants generate electricity through fuel combustion
Example 1:
Explain why a blue flame is preferred over a yellow flame.
▶️ Answer/Explanation
Blue flame indicates complete combustion
Produces more energy and less pollution
Conclusion: More efficient and cleaner.
Example 2:
State the conditions needed for combustion.
▶️ Answer/Explanation
Fuel, oxygen, and heat
Conclusion: All three are required for combustion.
Example 3 :
Explain why incomplete combustion is more likely in a poorly ventilated room.
▶️ Answer/Explanation
Step 1: Oxygen supply
Limited oxygen availability
Step 2: Reaction type
Leads to incomplete combustion
Step 3: Products
Produces carbon monoxide (CO)
Conclusion: Poor ventilation increases risk of toxic gas formation.