IB MYP Integrated Science- Chemistry- Corrosion and rusting-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Corrosion and rusting -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Corrosion and rusting -Study Notes -As per latest Syllabus.
Key Concepts:
Corrosion and rusting
IB MYP Integrated Science -Concise Summary Notes- All Topics
Corrosion and Rusting
Corrosion is the gradual destruction of metals due to chemical reactions with their environment. It usually involves oxidation and leads to the formation of compounds such as oxides.
Rusting
Rusting is a specific type of corrosion that occurs in iron and steel. It forms a reddish-brown substance called rust (hydrated iron(III) oxide).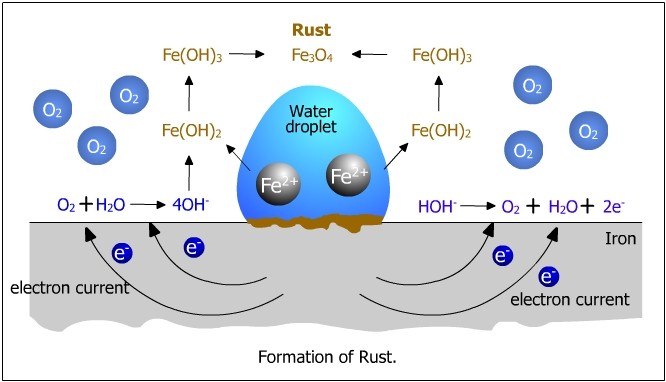
Conditions Required for Rusting
- Oxygen
- Water (moisture)
Both must be present for rusting to occur.
Rusting Reaction (Simplified)
\( \mathrm{Fe + O_2 + H_2O \rightarrow Fe_2O_3 \cdot xH_2O} \)
Process of Rusting (Redox Explanation)
- Iron is oxidised (loses electrons)
- Oxygen is reduced (gains electrons)
Factors Affecting Rusting
- Salt: speeds up rusting (e.g., seawater)
- Acidic conditions: increase corrosion rate
- Temperature: higher temperature increases rate
Prevention of Rusting
Rusting can be prevented by stopping contact with oxygen or water.

- Painting: forms a protective layer
- Oiling/greasing: prevents moisture contact
- Galvanising: coating with zinc
- Electroplating: coating with another metal
- Alloying: making stainless steel
Galvanising (Important Concept)
Zinc protects iron in two ways:
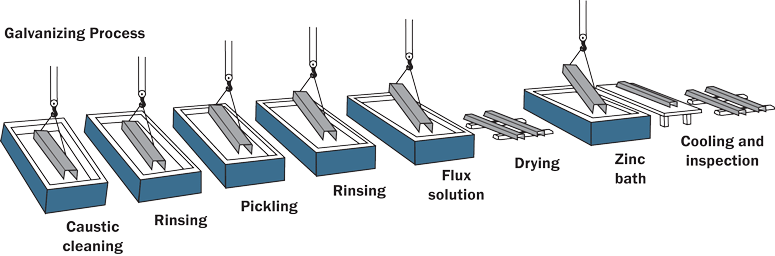
- Acts as a barrier
- Sacrificial protection (zinc corrodes instead of iron)
\( \mathrm{Zn \rightarrow Zn^{2+} + 2e^-} \)
Economic and Environmental Impact
- Damage to buildings, bridges, and vehicles
- High maintenance and replacement costs
- Wastage of materials and resources
Link to Reactivity Series
More reactive metals corrode more easily. Less reactive metals like gold do not corrode.
Real-World Applications
- Galvanised iron used in roofing
- Paint protects cars and bridges
- Stainless steel used in kitchen utensils
Example 1:
State the conditions necessary for rusting.
▶️ Answer/Explanation
Oxygen and water
Conclusion: Both are required for rusting.
Example 2:
Explain why salt water increases the rate of rusting.
▶️ Answer/Explanation
Salt increases electrical conductivity
Speeds up redox reactions
Conclusion: Rusting occurs faster.
Example 3 :
Explain how galvanising protects iron even if the coating is scratched.
▶️ Answer/Explanation
Step 1: Exposure
Iron becomes exposed
Step 2: Zinc reaction
Zinc oxidises instead of iron
Step 3: Protection
Zinc acts as sacrificial metal
Conclusion: Iron remains protected due to zinc’s higher reactivity.
