IB MYP Integrated Science- Chemistry- Diffusion and osmosis-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Diffusion and osmosis -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Diffusion and osmosis -Study Notes -As per latest Syllabus.
Key Concepts:
Diffusion and osmosis
IB MYP Integrated Science -Concise Summary Notes- All Topics
Diffusion and Osmosis
Diffusion and osmosis are processes that describe how particles move. They are explained using the particle model and kinetic theory, where particles are in constant random motion.
Diffusion
Diffusion is the net movement of particles from a region of higher concentration to a region of lower concentration due to random motion.

\( \mathrm{Diffusion:\ high\ concentration \rightarrow low\ concentration} \)
Key Features of Diffusion
- Occurs in gases, liquids, and (very slowly) in solids
- Does not require energy (passive process)
- Caused by random motion of particles
Factors Affecting Diffusion
- Temperature: higher temperature → faster diffusion
- Concentration gradient: greater difference → faster diffusion
- Particle size: smaller particles diffuse faster
Examples of Diffusion
- Perfume spreading in air
- Food colouring spreading in water
Osmosis
Osmosis is the movement of water molecules from a region of higher water concentration (dilute solution) to a region of lower water concentration (concentrated solution) through a partially permeable membrane.
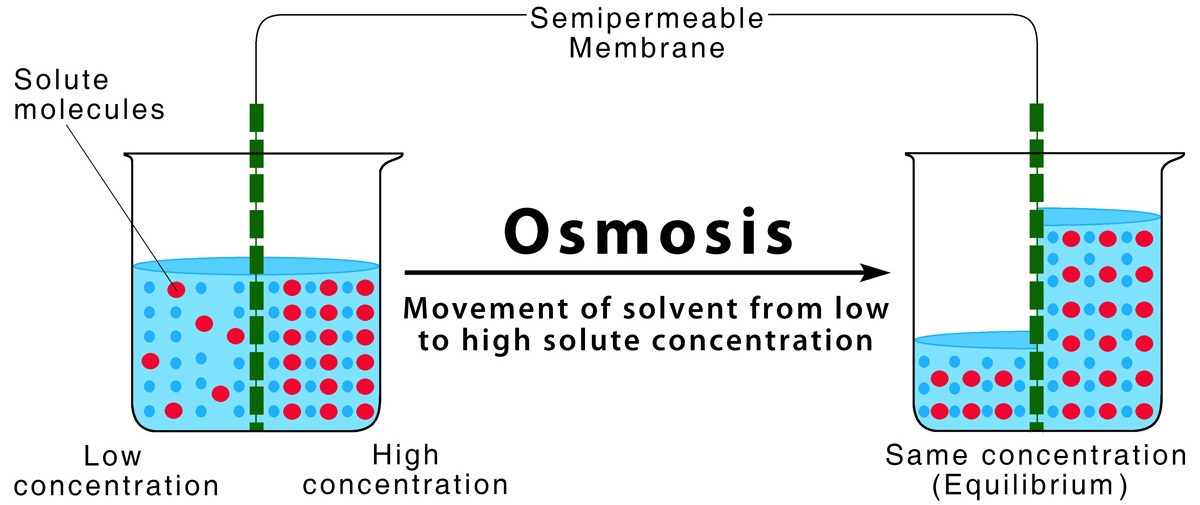
\( \mathrm{Osmosis:\ dilute \rightarrow concentrated\ solution} \)
Key Features of Osmosis
- Only involves water molecules
- Requires a partially permeable membrane
- Passive process (no energy required)
Terms Related to Osmosis
- Dilute solution: high water concentration
- Concentrated solution: low water concentration
Effects of Osmosis in Cells
- Hypotonic solution: water enters cell → cell swells
- Hypertonic solution: water leaves cell → cell shrinks
- Isotonic solution: no net movement of water
Diffusion vs Osmosis
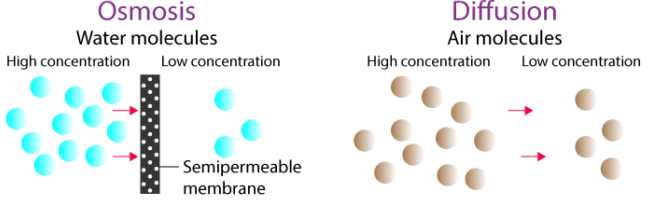
- Diffusion: movement of any particles
- Osmosis: movement of water only
- Osmosis requires a membrane; diffusion does not
Link to Kinetic Theory
Both diffusion and osmosis occur due to the random motion of particles, which is explained by kinetic theory.

Real-World Applications
- Gas exchange in lungs occurs by diffusion
- Water uptake in plant roots occurs by osmosis
- Food preservation (salt/sugar) uses osmosis
Example 1:
Explain why diffusion is faster at higher temperatures.
▶️ Answer/Explanation
Particles gain kinetic energy
Move faster → spread more quickly
Conclusion: Higher temperature increases diffusion rate.
Example 2:
Describe what happens to a plant cell in a hypotonic solution.
▶️ Answer/Explanation
Water enters the cell by osmosis
Cell swells and becomes turgid
Conclusion: Cell gains water and becomes firm.
Example 3 :
A cell is placed in a concentrated sugar solution.
Explain what happens using osmosis and kinetic theory.
▶️ Answer/Explanation
Step 1: Water concentration
Outside solution has lower water concentration
Step 2: Movement
Water moves out of the cell
Step 3: Result
Cell shrinks due to water loss
Conclusion: Osmosis causes water loss, explained by random particle motion.
