IB MYP Integrated Science- Chemistry- Electron configuration-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Electron configuration -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Electron configuration -Study Notes -As per latest Syllabus.
Key Concepts:
Electron configuration
IB MYP Integrated Science -Concise Summary Notes- All Topics
Electron Configuration
Electron configuration describes how electrons are arranged in energy levels (shells) around the nucleus of an atom. This arrangement determines the chemical properties and reactivity of an element.
Energy Levels (Shells)
Electrons occupy specific energy levels around the nucleus. These are sometimes called shells.
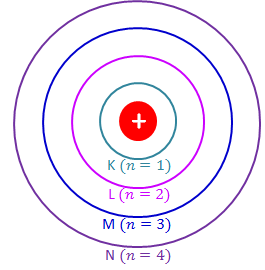
- First shell: maximum 2 electrons
- Second shell: maximum 8 electrons
- Third shell: maximum 8 electrons (for MYP level)
Filling Order (Electron Arrangement Rules)
Electrons fill energy levels starting from the lowest energy level first before moving to higher ones.
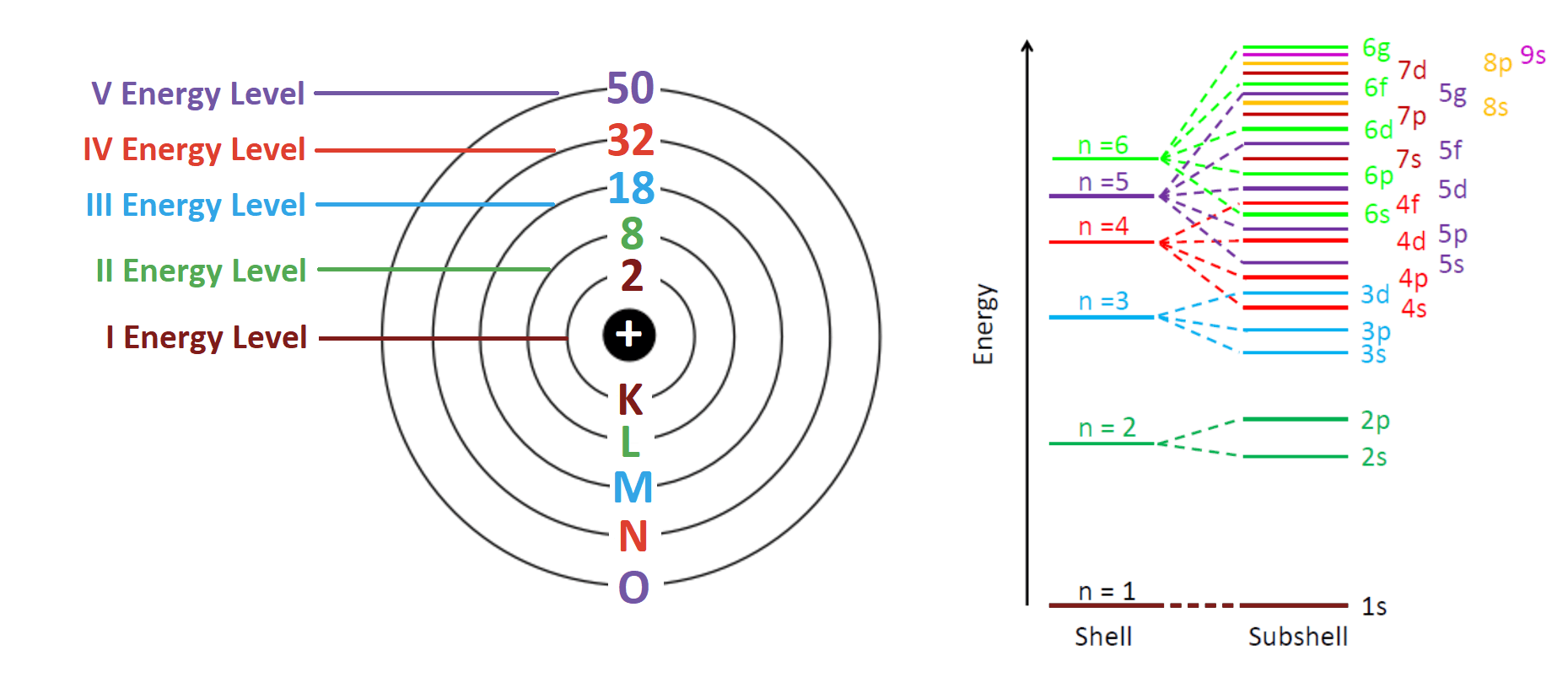
Key rules:
- Electrons fill the lowest energy level available first
- Each shell has a maximum capacity
- Outer shells determine chemical behavior
Writing Electron Configuration
Electron configuration is written as numbers showing how many electrons are in each shell.
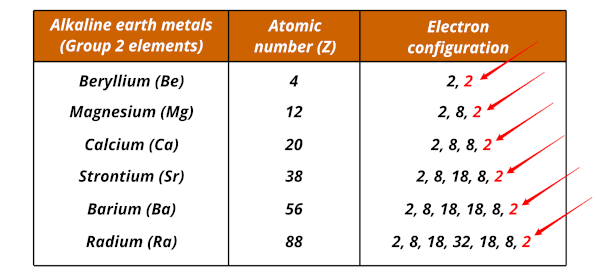
Examples:
- Helium (2): \( 2 \)
- Carbon (6): \( 2,4 \)
- Neon (10): \( 2,8 \)
- Magnesium (12): \( 2,8,2 \)
Valence Electrons and Stability
The outermost electrons are called valence electrons. Atoms tend to become more stable when their outer shell is full.
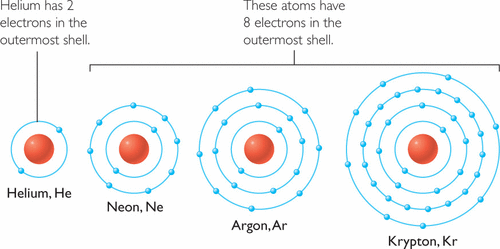
A full outer shell (usually 8 electrons) is called a stable electron configuration (noble gas configuration).
Formation of Ions (Link to Electron Configuration)
Atoms can gain or lose electrons to achieve a full outer shell.
- Metals tend to lose electrons → form positive ions
- Non-metals tend to gain electrons → form negative ions
Example:
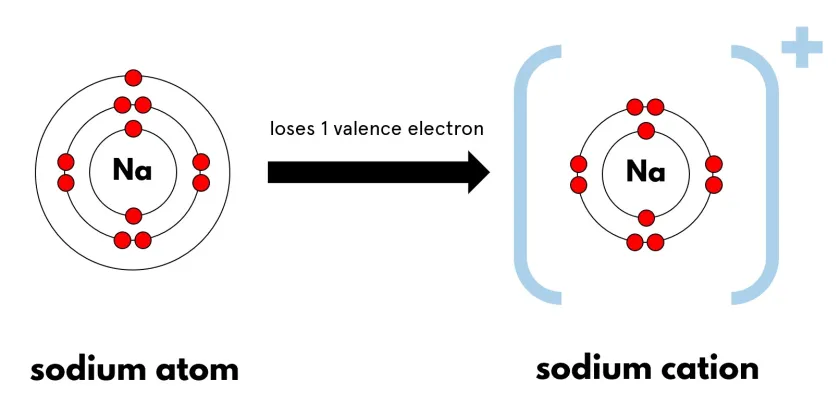
\( \mathrm{Na: 2,8,1 \rightarrow Na^+: 2,8} \)
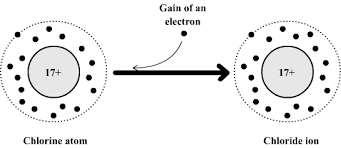
\( \mathrm{Cl: 2,8,7 \rightarrow Cl^-: 2,8,8} \)
Bohr Diagrams
Electron configuration can be represented visually using Bohr diagrams, where electrons are shown in circular shells around the nucleus.
Real-World Importance
Electron configuration explains why elements react, form bonds, and create compounds essential for life.
Example 1:
Write the electron configuration of an atom with atomic number 15.
▶️ Answer/Explanation
Total electrons = 15
Fill shells: \( 2,8,5 \)
Conclusion: Electron configuration is \( 2,8,5 \).
Example 2:
An element has the electron configuration \( 2,8,2 \).
a) Identify the element
b) State its group and period
▶️ Answer/Explanation
Step 1: Total electrons
\( 2 + 8 + 2 = 12 \)
Step 2: Identify element
Atomic number 12 → Magnesium (Mg)
Step 3: Group and Period
Group = 2 (valence electrons)
Period = 3 (number of shells)
Conclusion: Magnesium, Group 2, Period 3.
Example 3:
An atom forms an ion \( \mathrm{X^{2-}} \) with electron configuration \( 2,8,8 \).
a) Determine the electron configuration of the neutral atom
b) Identify the element
c) Explain why the ion is formed
▶️ Answer/Explanation
Step 1: Electrons in ion
\( 2 + 8 + 8 = 18 \)
Step 2: Neutral atom electrons
\( 18 – 2 = 16 \)
Step 3: Electron configuration
\( 2,8,6 \)
Step 4: Identify element
Atomic number 16 → Sulfur (S)
Step 5: Explanation
The atom gains 2 electrons to achieve a full outer shell \( (2,8,8) \), which is a stable noble gas configuration.
Conclusion: Neutral atom is sulfur with configuration \( 2,8,6 \), forming \( \mathrm{S^{2-}} \) for stability.
