IB MYP Integrated Science- Chemistry- Environmental implications-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Environmental implications -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Environmental implications -Study Notes -As per latest Syllabus.
Key Concepts:
Environmental implications
IB MYP Integrated Science -Concise Summary Notes- All Topics
Environmental Implications
Chemical processes such as combustion, extraction of metals, and industrial reactions can have significant impacts on the environment. Understanding these effects helps in developing sustainable solutions.
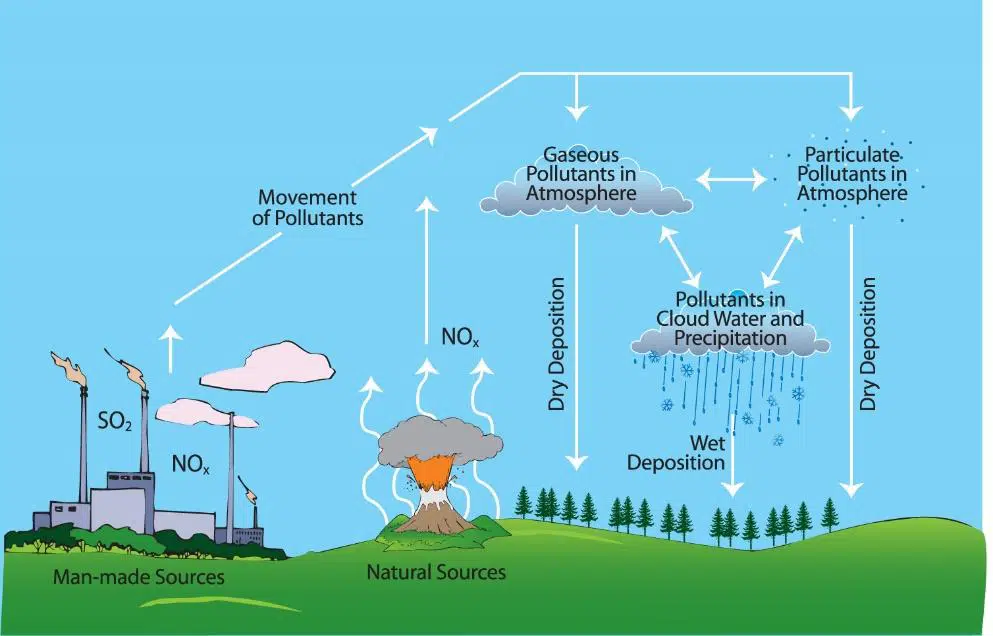
Air Pollution
Air pollution is caused by harmful gases and particles released into the atmosphere.

- Carbon dioxide \( \mathrm{CO_2} \): contributes to global warming
- Carbon monoxide \( \mathrm{CO} \): toxic gas from incomplete combustion
- Sulfur dioxide \( \mathrm{SO_2} \): causes acid rain
- Nitrogen oxides \( \mathrm{NO_x} \): contribute to smog and acid rain
- Particulates (soot): cause respiratory problems
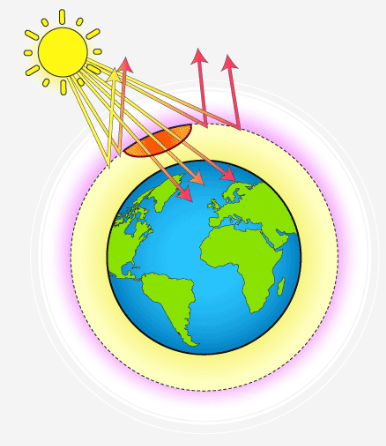
Greenhouse Effect and Global Warming
Greenhouse gases trap heat in the atmosphere, leading to an increase in Earth’s temperature.
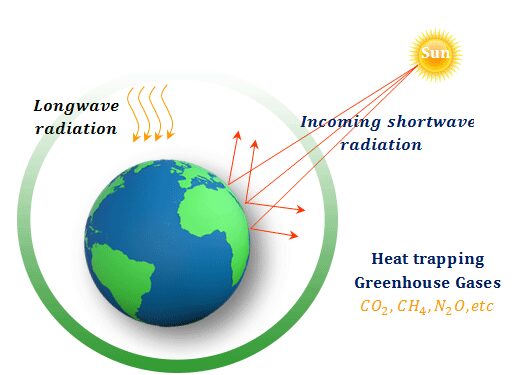
\( \mathrm{CO_2 + heat \rightarrow trapped\ energy} \)
- Main gases: \( \mathrm{CO_2} \), methane \( \mathrm{CH_4} \)
- Causes climate change, rising sea levels
Acid Rain
Acid rain forms when sulfur dioxide and nitrogen oxides react with water in the atmosphere.
\( \mathrm{SO_2 + H_2O \rightarrow H_2SO_4} \)
- Damages buildings and monuments
- Harms plants and aquatic life
Water Pollution

- Industrial waste contaminates rivers and lakes
- Fertilisers cause eutrophication (excess algae growth)
- Oil spills harm marine life
Land Pollution

- Mining damages landscapes
- Waste disposal leads to soil contamination
- Landfills produce methane gas
Ozone Layer Depletion
Certain chemicals (e.g., CFCs) break down ozone in the upper atmosphere.
- Ozone protects Earth from harmful UV radiation
- Depletion increases risk of skin cancer
Sustainable Solutions
- Use renewable energy sources
- Reduce, reuse, recycle materials
- Install filters and catalytic converters
- Develop cleaner technologies
Link to Chemistry Topics
- Combustion → produces greenhouse gases
- Extraction → environmental damage from mining
- Acids and bases → acid rain effects
Real-World Importance
- Protecting ecosystems and biodiversity
- Reducing climate change impacts
- Ensuring sustainable use of resources
Example 1:
Explain how combustion of fossil fuels contributes to global warming.
▶️ Answer/Explanation
Combustion releases \( \mathrm{CO_2} \)
\( \mathrm{CO_2} \) traps heat in atmosphere
Conclusion: Leads to increase in global temperatures.
Example 2:
Describe the effects of acid rain on the environment.
▶️ Answer/Explanation
Damages buildings
Harms plants and aquatic organisms
Conclusion: Acid rain has harmful environmental effects.
Example 3:
Explain how catalytic converters reduce air pollution from vehicles.
▶️ Answer/Explanation
Step 1: Harmful gases
CO and NOx produced in engines
Step 2: Conversion
CO → \( \mathrm{CO_2} \)
NOx → \( \mathrm{N_2} \)
Conclusion: Harmful gases are converted into less harmful substances.
