IB MYP Integrated Science- Chemistry- Extraction-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Extraction -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Extraction -Study Notes -As per latest Syllabus.
Key Concepts:
Extraction
IB MYP Integrated Science -Concise Summary Notes- All Topics
Extraction of Metals
Extraction is the process of removing a metal from its ore. Most metals are found in nature as compounds (ores), and chemical processes are required to obtain the pure metal.
What is an Ore?

An ore is a naturally occurring rock that contains enough metal to make extraction economically worthwhile.
Methods of Extraction
The method used depends on the reactivity of the metal.
1. Extraction by Reduction with Carbon
Metals that are less reactive than carbon can be extracted by heating with carbon.
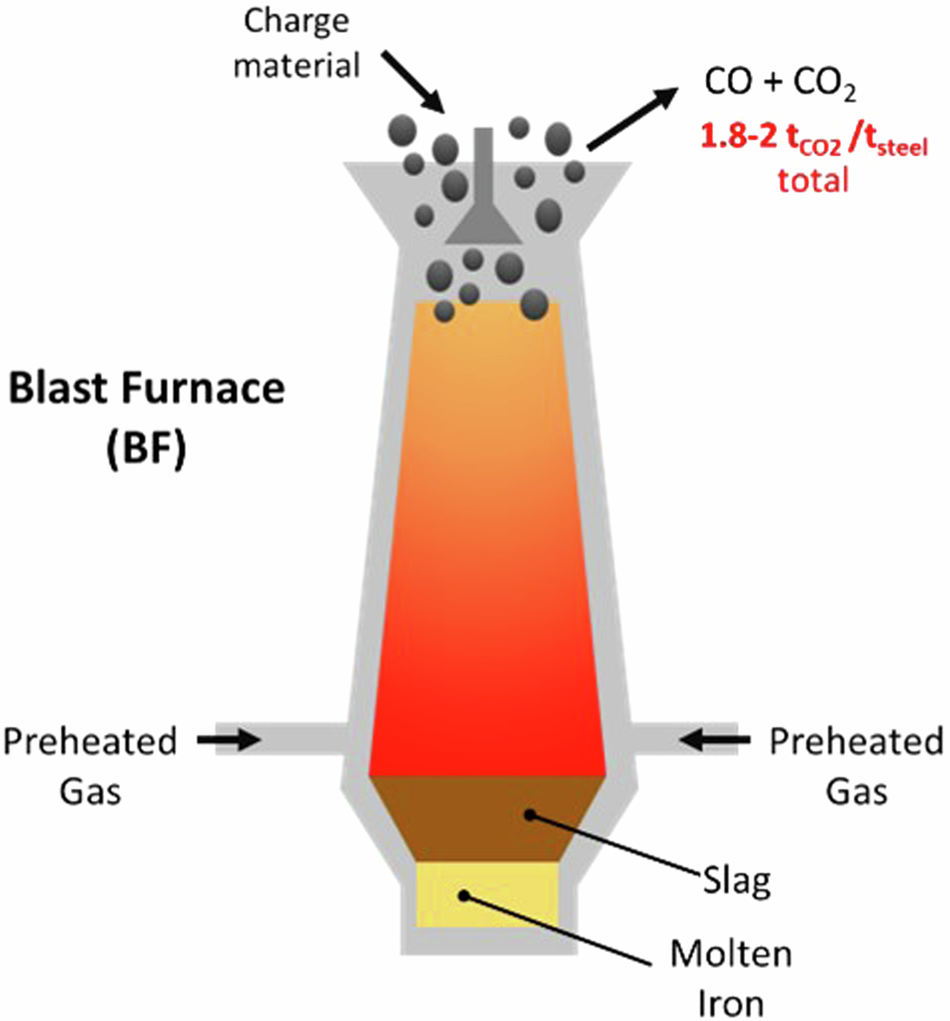
Example: Iron extraction
\( \mathrm{Fe_2O_3 + 3CO \rightarrow 2Fe + 3CO_2} \)
Carbon monoxide removes oxygen from iron oxide (reduction).
2. Extraction by Electrolysis
Metals more reactive than carbon (e.g., aluminium) are extracted using electrolysis.
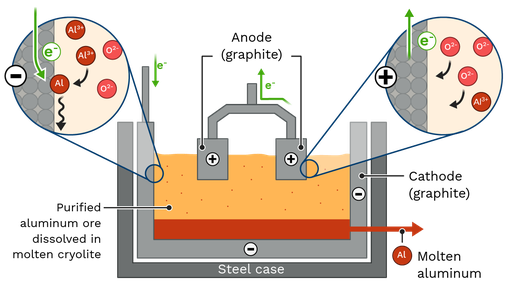
Example: Aluminium extraction
\( \mathrm{Al_2O_3 \rightarrow 2Al + \frac{3}{2}O_2} \)
Electric current is used to break down the compound.
3. Native Metals
Very unreactive metals (e.g., gold, silver) are found as pure elements and require minimal extraction.
The Reactivity Series
The reactivity series helps predict extraction methods:
- Highly reactive metals → electrolysis
- Moderately reactive → reduction with carbon
- Unreactive → found native
Reduction and Oxidation
- Reduction: loss of oxygen or gain of electrons
- Oxidation: gain of oxygen or loss of electrons
Environmental Considerations
- Mining causes land damage
- Carbon extraction produces \( \mathrm{CO_2} \)
- Electrolysis requires large amounts of energy
Recycling Metals
Recycling metals reduces the need for extraction, saves energy, and reduces environmental impact.
Real-World Applications
- Iron is used in construction
- Aluminium is used in aircraft and packaging
- Copper is used in electrical wiring
Example 1:
Explain why aluminium is extracted by electrolysis and not by carbon reduction.
▶️ Answer/Explanation
Aluminium is more reactive than carbon
Carbon cannot remove oxygen from aluminium oxide
Conclusion: Electrolysis is required.
Example 2:
Describe how iron is extracted from its ore.
▶️ Answer/Explanation
Iron oxide is heated with carbon monoxide
Reduction occurs to form iron
\( \mathrm{Fe_2O_3 + 3CO \rightarrow 2Fe + 3CO_2} \)
Conclusion: Iron is extracted by reduction.
Example 3:
A metal oxide is heated with carbon but no reaction occurs.
Explain what this indicates about the metal and suggest a method of extraction.
▶️ Answer/Explanation
Step 1: Observation
No reduction by carbon
Step 2: Interpretation
Metal is more reactive than carbon
Step 3: Method
Use electrolysis
Conclusion: Highly reactive metals require electrolysis for extraction.
