IB MYP Integrated Science- Chemistry- Nuclear energy-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Nuclear energy -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Nuclear energy -Study Notes -As per latest Syllabus.
Key Concepts:
Nuclear energy
IB MYP Integrated Science -Concise Summary Notes- All Topics
Nuclear Chemistry and Nuclear Energy
Nuclear chemistry involves changes in the nucleus of an atom. These changes release large amounts of energy, much greater than chemical reactions.
Atomic Structure![]()
The nucleus contains protons and neutrons, and nuclear reactions involve changes in these particles.
Radioactivity
Radioactivity is the spontaneous emission of radiation from unstable nuclei.
Types of Nuclear Radiation
- Alpha (α): helium nucleus \( \mathrm{^{4}_{2}He} \)
- Beta (β): electron \( \mathrm{^{0}_{-1}e} \)
- Gamma (γ): electromagnetic radiation
Properties of Radiation
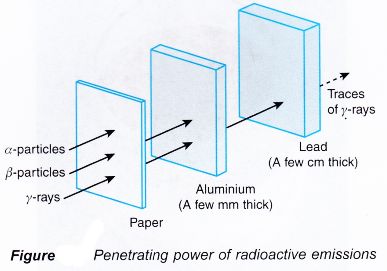
- Alpha: low penetration, highly ionising
- Beta: medium penetration
- Gamma: high penetration, low ionisation
Nuclear Reactions
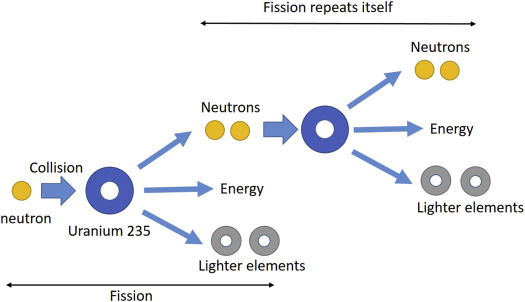
1. Nuclear Fission
A heavy nucleus splits into smaller nuclei, releasing energy.
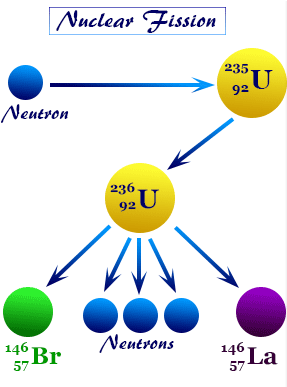
\( \mathrm{^{235}_{92}U + n \rightarrow ^{141}_{56}Ba + ^{92}_{36}Kr + 3n + energy} \)
- Used in nuclear power plants
- Produces chain reactions
2. Nuclear Fusion
Two light nuclei combine to form a heavier nucleus, releasing energy.
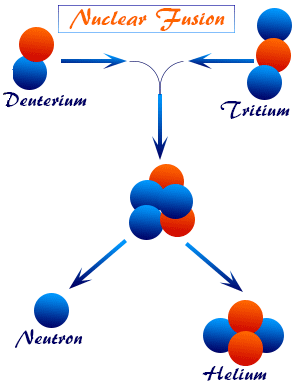
\( \mathrm{^{2}_{1}H + ^{3}_{1}H \rightarrow ^{4}_{2}He + n + energy} \)
- Occurs in the Sun
- Releases more energy than fission
Nuclear Energy
Nuclear energy is the energy released during nuclear reactions (fission or fusion).
Advantages of Nuclear Energy
| Disadvantages of Nuclear Energy
| Applications of Nuclear Chemistry
| Safety Measures
|
Environmental Impact
- Low air pollution compared to fossil fuels
- Long-term radioactive waste hazards
Example 1:
State one difference between alpha and gamma radiation.
▶️ Answer/Explanation
Alpha has low penetration, gamma has high penetration
Conclusion: Gamma is more penetrating than alpha.
Example 2:
Explain why nuclear energy releases more energy than chemical reactions.
▶️ Answer/Explanation
Energy comes from nucleus, not electron rearrangement
Stronger nuclear forces involved
Conclusion: Nuclear changes release much more energy.
Example 3 :
A radioactive isotope has a half-life of 10 years. If the initial amount is 80 g, calculate the amount remaining after 30 years.
▶️ Answer/Explanation
Step 1: Number of half-lives
\( 30 \div 10 = 3 \)
Step 2: Calculate remaining amount
\( 80 \rightarrow 40 \rightarrow 20 \rightarrow 10 \)
Conclusion: 10 g remains after 30 years.
