IB MYP Integrated Science- Chemistry- Particles and kinetic theory-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Particles and kinetic theory -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Particles and kinetic theory -Study Notes -As per latest Syllabus.
Key Concepts:
Particles and kinetic theory
IB MYP Integrated Science -Concise Summary Notes- All Topics
Particle Model and Kinetic Theory
The particle model explains that all matter is made of tiny particles (atoms or molecules). The kinetic theory builds on this by explaining that these particles are always moving and that their motion (kinetic energy) determines the physical properties of matter.
Key Principles of Kinetic Theory

- All matter is made of tiny particles
- Particles are in constant random motion
- Particles possess kinetic energy
- Temperature is proportional to average kinetic energy
Kinetic Energy and Temperature
As temperature increases, particles gain kinetic energy and move faster.
\( \mathrm{Temperature \propto \text{average kinetic energy}} \)
This explains why heating a substance causes expansion, faster diffusion, and changes of state.
States of Matter Explained by Kinetic Theory
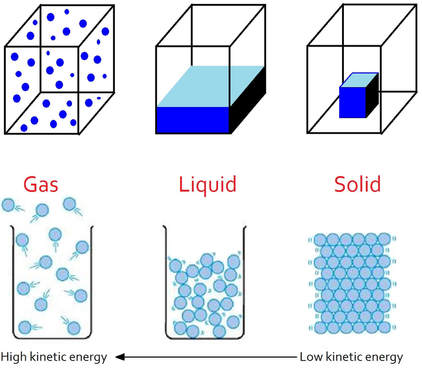
Solids
| Liquids
| Gases
|
Changes of State (Energy Perspective)
Changes of state occur when energy is transferred to or from particles.
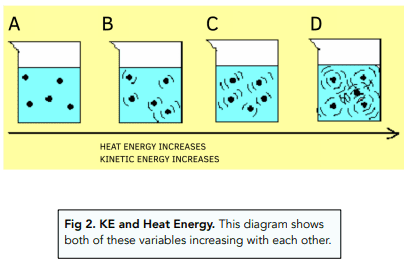
- Heating → increases kinetic energy
- Cooling → decreases kinetic energy
During a change of state, energy is used to overcome forces between particles rather than increase temperature.
Gas Pressure (Kinetic Theory)
Gas pressure is caused by particles colliding with the walls of a container.
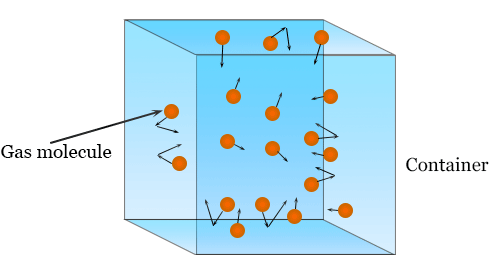
Higher kinetic energy leads to:
- More frequent collisions
- Greater force per collision
Relationship Summary
\( \mathrm{Higher\ temperature \rightarrow higher\ kinetic\ energy \rightarrow faster\ particles} \)
Real-World Applications
- Perfume spreading is due to diffusion
- Heating gases increases pressure (e.g., tyres)
- Cooking involves increased particle motion
Example 1:
Explain why increasing temperature increases the speed of gas particles.
▶️ Answer/Explanation
Temperature increases kinetic energy
Higher kinetic energy → faster motion
Conclusion: Particle speed increases with temperature.
Example 2:
Explain why gas pressure increases when temperature increases.
▶️ Answer/Explanation
Particles move faster
Collisions with walls increase
Collisions are more forceful
Conclusion: Pressure increases due to increased collision frequency and force.
Example 3:
A gas is cooled at constant pressure.
Explain what happens to its volume using kinetic theory.
▶️ Answer/Explanation
Step 1: Energy decreases
Particles lose kinetic energy
Step 2: Movement slows
Particles move more slowly
Step 3: Volume change
Particles come closer together → volume decreases
Conclusion: Cooling reduces kinetic energy, causing gas volume to decrease.