IB MYP Integrated Science- Chemistry- pH scale and indicators-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – pH scale and indicators -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – pH scale and indicators -Study Notes -As per latest Syllabus.
Key Concepts:
pH scale and indicators
IB MYP Integrated Science -Concise Summary Notes- All Topics
pH Scale and Indicators
The pH scale is used to measure how acidic or alkaline (basic) a substance is. It is based on the concentration of hydrogen ions \( \mathrm{H^+} \) present in a solution.
The pH Scale
The pH scale ranges from 0 to 14:
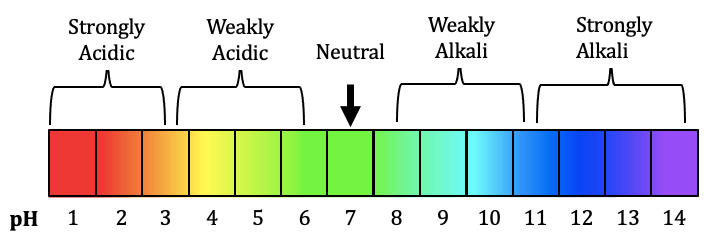
- pH 0–6 → acidic
- pH 7 → neutral
- pH 8–14 → alkaline (basic)
The lower the pH, the higher the concentration of \( \mathrm{H^+} \) ions, and the more acidic the solution is.
Nature of the pH Scale
The pH scale is logarithmic, meaning each change of 1 pH unit represents a tenfold change in acidity.
\( \mathrm{pH = -\log[H^+]} \)
For example, a solution with pH 3 is 10 times more acidic than pH 4 and 100 times more acidic than pH 5.
Strong and Weak Acids/Bases (Link to pH)
- Strong acids → very low pH (0–3)
- Weak acids → higher pH (4–6)
- Weak bases → pH (8–10)
- Strong bases → high pH (11–14)
Indicators
Indicators are substances that change colour depending on the pH of a solution. They are used to identify whether a substance is acidic, neutral, or alkaline.
Common Indicators
Litmus
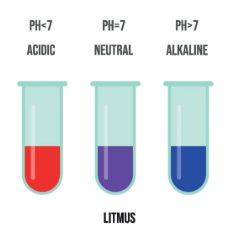
- Acid → red
- Base → blue
Universal Indicator
Universal indicator shows a range of colours depending on pH:
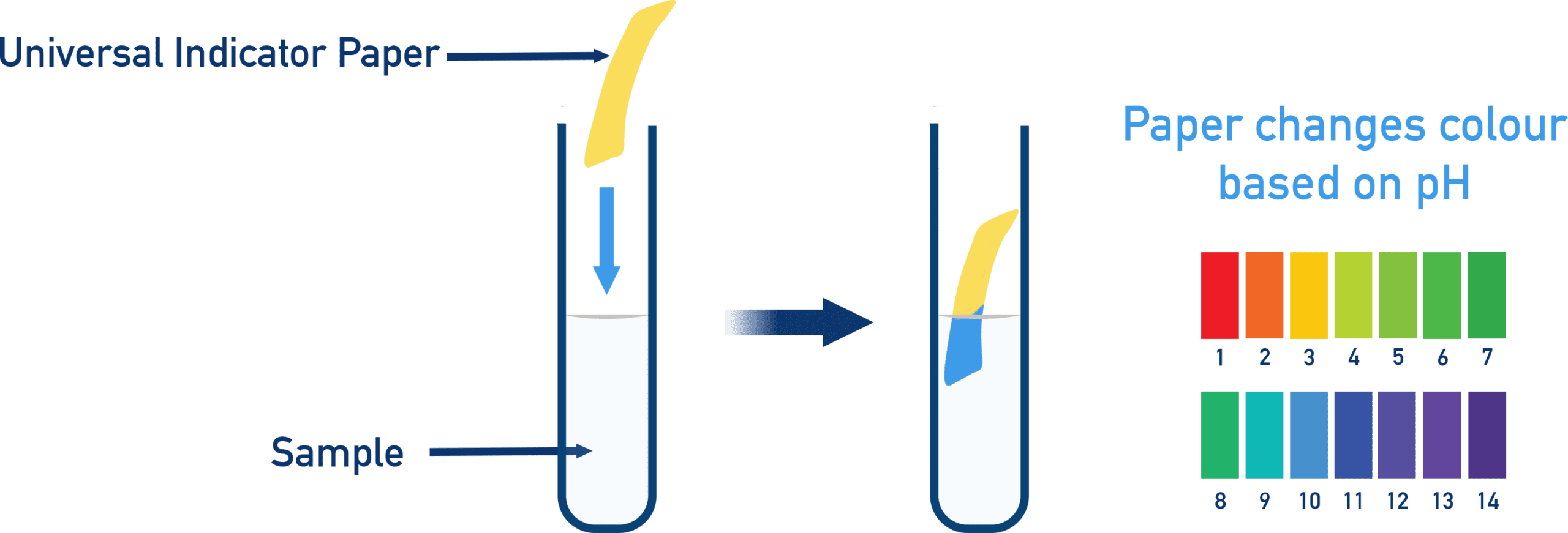
- Red → strong acid
- Orange/Yellow → weak acid
- Green → neutral
- Blue → weak base
- Purple → strong base
Other Indicators
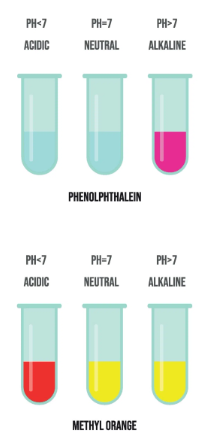
- Phenolphthalein: colourless (acid) → pink (base)
- Methyl orange: red (acid) → yellow (base)
Using Indicators
Indicators can be used to:
- Identify acids and bases
- Estimate pH
- Detect neutralisation reactions
Neutralisation and pH
When an acid reacts with a base, the pH moves toward 7.
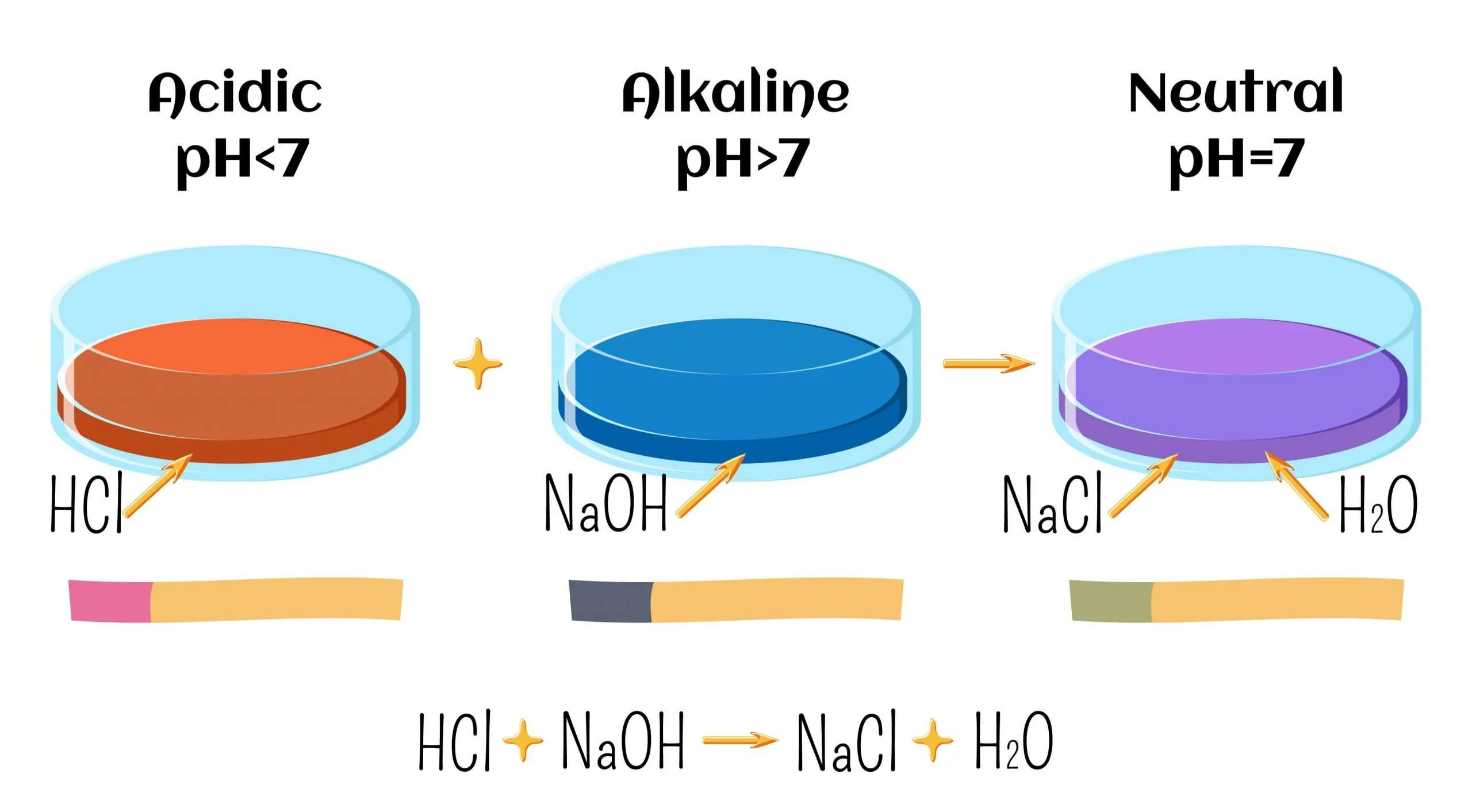
\( \mathrm{Acid + Base \rightarrow Salt + Water} \)
At the equivalence point, the solution is neutral (pH ≈ 7 for strong acid–strong base).
Real-World Applications
- pH of soil affects plant growth
- pH of blood must be maintained (~7.4)
- Water treatment uses pH control
Example 1:
A solution turns universal indicator red. What can you conclude about its pH?
▶️ Answer/Explanation
Red indicates strong acidity
pH is approximately between 0 and 3
Conclusion: The solution is strongly acidic.
Example 2:
Compare a solution of pH 2 and pH 5.
▶️ Answer/Explanation
pH 2 is more acidic than pH 5
Difference = 3 units → \( 10^3 = 1000 \)
pH 2 is 1000 times more acidic
Conclusion: Small pH changes represent large differences in acidity.
Example 3 :
A student performs a neutralisation reaction between hydrochloric acid and sodium hydroxide using phenolphthalein.
Explain the colour changes observed during the reaction.
▶️ Answer/Explanation
Step 1: Initial condition
Phenolphthalein is colourless in acidic solution
Step 2: Adding base
pH increases as base is added
Step 3: Endpoint
At neutralisation, solution becomes slightly basic → turns pink
Conclusion: Colour change indicates the endpoint of neutralisation.
