IB MYP Integrated Science- Chemistry- Properties of acids and bases-Study Notes - New Syllabus
IB MYP Integrated Science- Chemistry – Properties of acids and bases -Study Notes – New syllabus
IB MYP Integrated Science- Chemistry – Properties of acids and bases -Study Notes -As per latest Syllabus.
Key Concepts:
Properties of acids and bases
IB MYP Integrated Science -Concise Summary Notes- All Topics
Acids, Bases and Their Properties
Acids and bases are important classes of substances that are commonly found in everyday life and play key roles in chemical reactions. They can be identified by their properties and how they behave in aqueous solutions.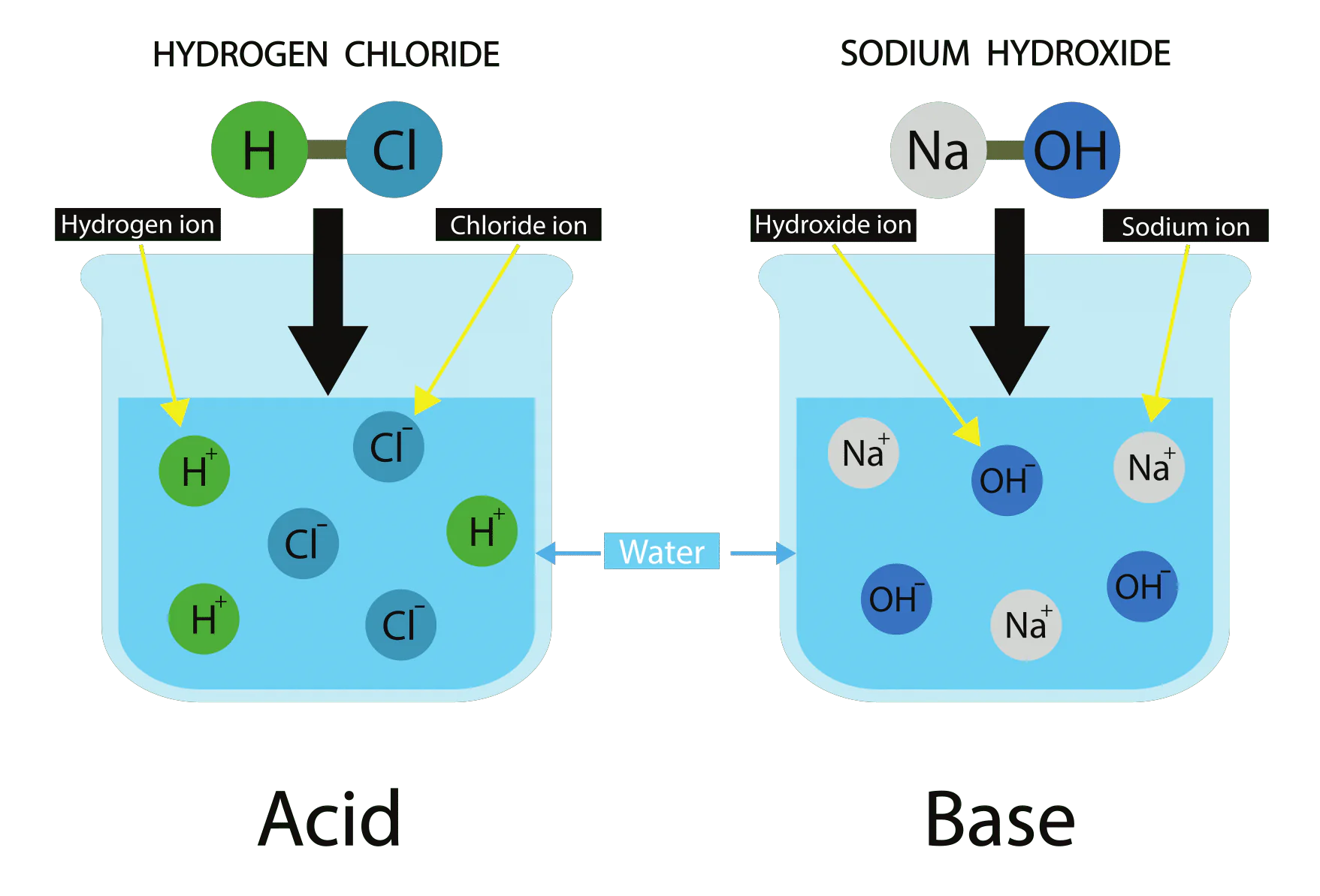
Definition of Acids and Bases
According to simple (Arrhenius) definitions:
- Acids produce hydrogen ions \( \mathrm{H^+} \) in aqueous solution
- Bases produce hydroxide ions \( \mathrm{OH^-} \) in aqueous solution
A alkali is a base that is soluble in water.
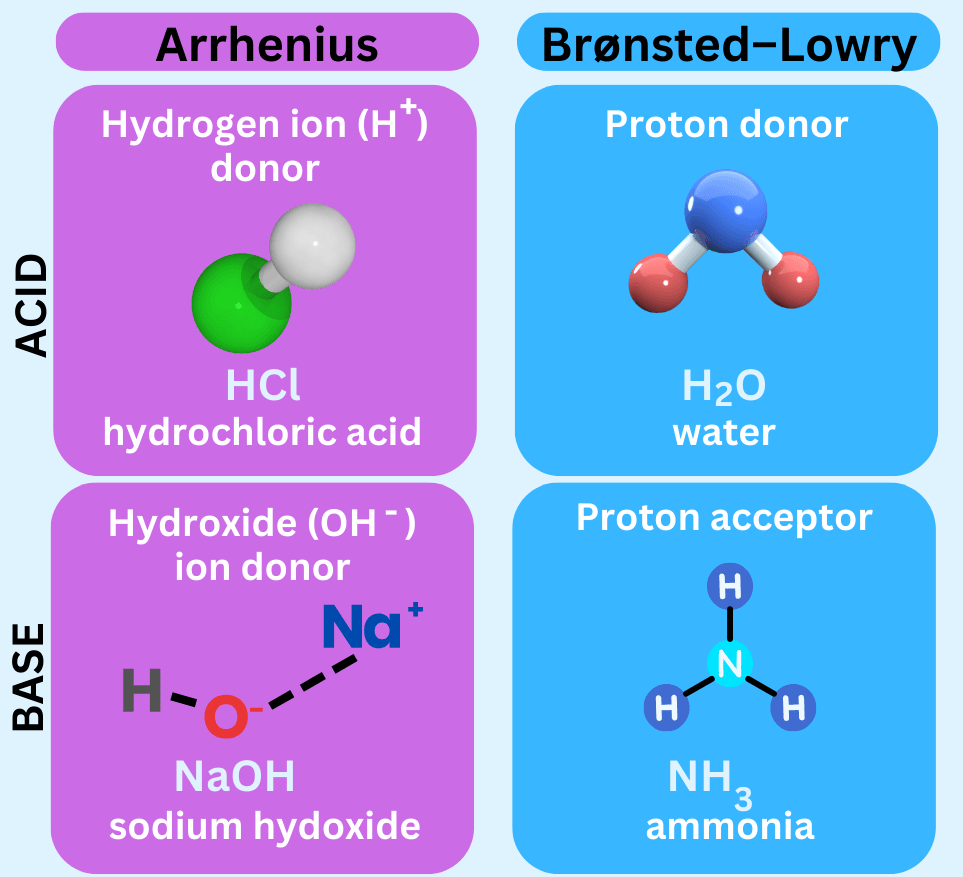
Examples of Acids and Bases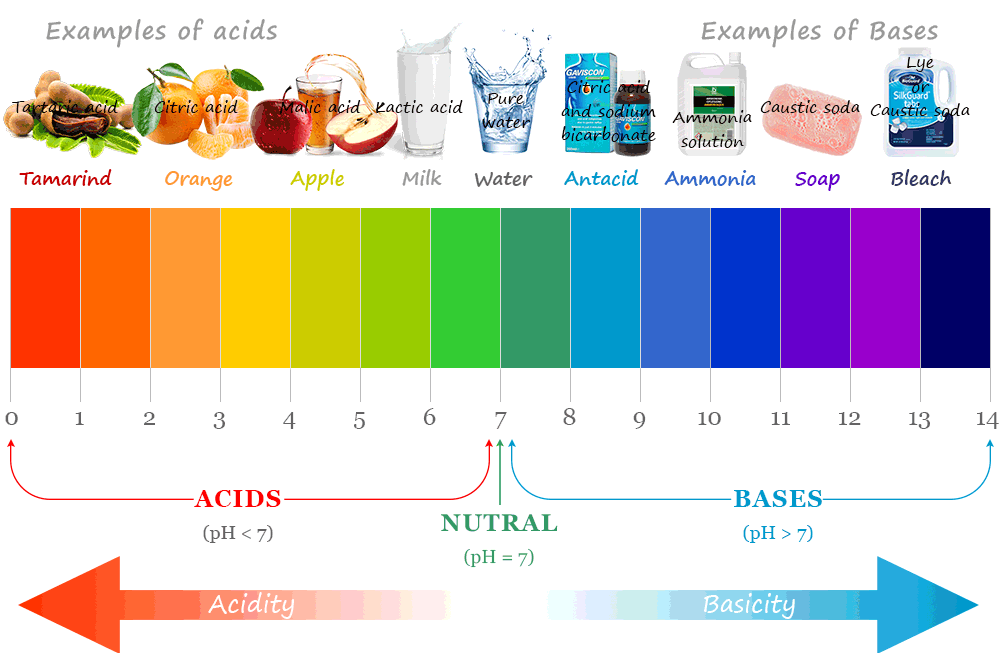
- \( \mathrm{HCl} \) (hydrochloric acid)
- \( \mathrm{H_2SO_4} \) (sulfuric acid)
- \( \mathrm{NaOH} \) (sodium hydroxide)
- \( \mathrm{KOH} \) (potassium hydroxide)
The pH Scale
The pH scale measures how acidic or alkaline a solution is.
- pH 0–6 → acidic
- pH 7 → neutral
- pH 8–14 → alkaline
Properties of Acids
| Properties of Bases / Alkalis
|
Lower pH means higher concentration of \( \mathrm{H^+} \) ions.
Strength of Acids and Bases
Strength refers to how completely a substance ionises in water.
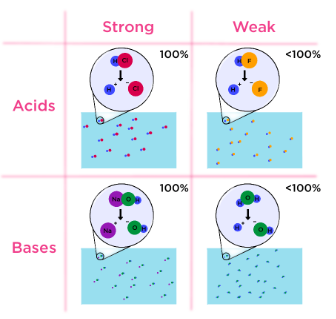
- Strong acids completely ionise (e.g., \( \mathrm{HCl} \))
- Weak acids partially ionise (e.g., \( \mathrm{CH_3COOH} \))
Neutralisation Reactions
When an acid reacts with a base, they neutralise each other to form salt and water.
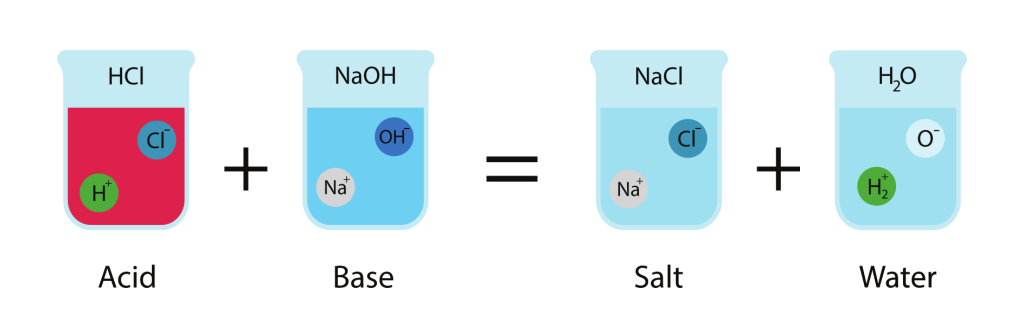
\( \mathrm{Acid + Base \rightarrow Salt + Water} \)
Example:
\( \mathrm{HCl + NaOH \rightarrow NaCl + H_2O} \)
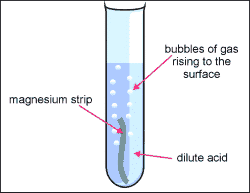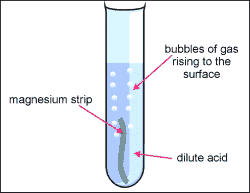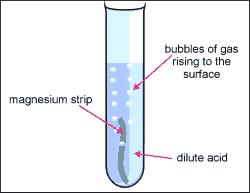
Acid Reactions
Acids react in characteristic ways:

- With metals → salt + hydrogen
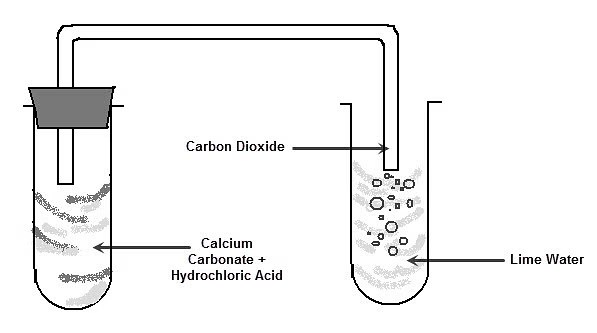
- With carbonates → salt + water + carbon dioxide
Examples:
\( \mathrm{Mg + 2HCl \rightarrow MgCl_2 + H_2} \)
\( \mathrm{CaCO_3 + 2HCl \rightarrow CaCl_2 + CO_2 + H_2O} \)
Base Reactions
Bases (alkalis) also react in characteristic ways:
- With acids → salt + water (neutralisation)
- With ammonium salts → ammonia gas + water + salt
Examples:
\( \mathrm{NaOH + HCl \rightarrow NaCl + H_2O} \)
\( \mathrm{NaOH + NH_4Cl \rightarrow NH_3 + H_2O + NaCl} \)
Real-World Applications
- Antacids neutralise excess stomach acid
- Soil pH is controlled in agriculture
- Acids are used in cleaning and industry
Example 1:
Describe what happens when hydrochloric acid reacts with magnesium.
▶️ Answer/Explanation
Magnesium reacts to form magnesium chloride and hydrogen gas
\( \mathrm{Mg + 2HCl \rightarrow MgCl_2 + H_2} \)
Conclusion: Acid + metal produces salt and hydrogen gas.
Example 2:
A solution has pH 3. Describe its properties.
▶️ Answer/Explanation
It is strongly acidic
High concentration of \( \mathrm{H^+} \)
Turns blue litmus red
Conclusion: Strong acidic solution.
Example 3 :
A student adds sodium carbonate to hydrochloric acid.
a) Predict the products
b) Write the balanced equation
c) Describe observations
▶️ Answer/Explanation
Step 1: Products
Salt (NaCl), water, carbon dioxide
Step 2: Equation
\( \mathrm{Na_2CO_3 + 2HCl \rightarrow 2NaCl + CO_2 + H_2O} \)
Step 3: Observations
Effervescence (bubbling) due to CO₂ gas
Conclusion: Acid + carbonate reaction produces gas and fizzing.